Is Momelotinib a domestic or imported drug manufacturer?
Momelotinib is a JAK1/JAK2 inhibitor that is mainly used to treat patients with moderate-to-high-risk myelofibrosis (MF) with symptoms of anemia, including primary myelofibrosis and myelofibrosis secondary to polycythemia vera or essential thrombocythemia. Its mechanism of action not only targets disease-related splenomegaly and systemic symptoms, but also improves patients' anemia, which gives it certain differentiated advantages among similar drugs.
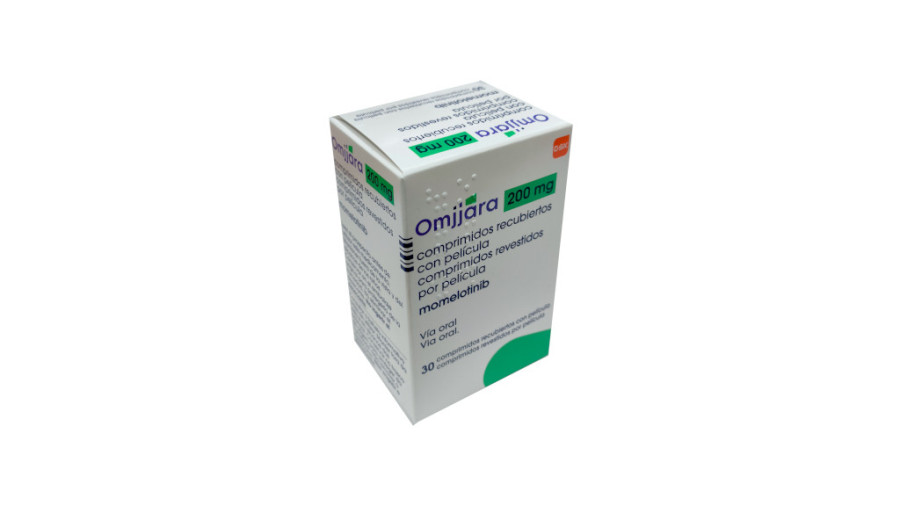
Currently, the original drug of molotinib is developed and produced by the American biopharmaceutical companySierra Oncology (later acquired by GSK) and is an imported drug. In the Chinese market, as of now, there is no domestic version on the market, and there is no clear local manufacturer. If domestic doctors want to use the drug, they usually need to obtain it through overseas drug purchase channels, including the US version and the specifications listed in other countries. Common packaging specifications in overseas markets include 100mg 30 tablets, 150mg 30 tablets and 200mg*30 tablets, allowing doctors to choose the appropriate dosage according to the patient’s specific conditions.
In terms of cross-border purchases, patients need to pay attention to the legal and compliant import channels of drugs and avoid purchasing products whose quality cannot be guaranteed through unknown channels. According to regulations, there are usually certain policies for individuals to bring small amounts of prescription drugs for self-use back to the country, especially prescription drugs that have not been registered domestically. Therefore, they must obtain them under the guidance of a doctor and through a qualified international drug platform or an international pharmacy of a formal medical institution to ensure that the source of the drugs is formal and the transportation and storage conditions meet the requirements.
Generally speaking, molotinib is currently an imported original drug and has no domestic market record. If patients need the drug in the short term, they need to rely on overseas channels. In the future, as pharmaceutical companies advance registration and policy approvals accelerate, the possibility of the drug entering the Chinese market and being included in medical insurance cannot be ruled out, which will provide more patients with myelofibrosis with more accessible treatment options.
Reference materials:https://en.wikipedia.org/wiki/Momelotinib
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















