Where can I buy nirogacestat?
Nirogacestat is a new gamma secretase inhibitor developed by the American pharmaceutical company SpringWorks Therapeutics. Its main indication is the treatment of adult patients with progressive desmoid tumors. Desmoid tumor is a rare but aggressive benign tumor that often grows in soft tissue and invades surrounding structures. Although it does not have the ability to metastasize, it is locally destructive and has a high recurrence rate, which has a significant impact on patients' quality of life. Nilogastat brings new treatment options to these patients, especially those who require systemic therapy but have poor response to traditional surgery, radiation therapy or drugs.
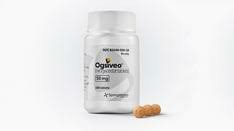
As of now, nirogalastat has not been officially launched in mainland China, so the drug cannot be obtained through regular prescription channels in domestic hospital pharmacies. Because of this, its price has not yet entered the national medical insurance system, and no formal reimbursement ratio or clinical application guidelines have been announced. According to overseas information, Nilogastat has been approved by the FDA in the United States and is sold under the trade name Ogsiveo®. The specification is 50 mg × 180 tablets/box, and the price per box is as high as approximately 280,000 yuan. It is a high-priced innovative rare disease drug. There are currently no legal generic drugs on the market, which means patients almost have to rely on the supply of original drugs.
For domestic patients, if they urgently need to use Nilogastat, they can try to obtain it through the following two methods: First, international medical referral, through medical institutions with overseas medical channels, go to the United States and other countries where the drug is listed for treatment or issue prescription drugs and mail them back to the country; second, cross-border drug purchase through formal overseas pharmacy authorization platforms, but patients should focus on screening whether the source of the drug is legal and use the drug under the guidance of a professional physician. In addition, some rare disease public welfare platforms or patient assistance projects may also help patients apply for clinical trials or compassionate use channels for the drug, which deserves attention.
Reference materials:https://www.drugs.com/mtm/nirogacestat.html
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















