Is selumetinib worth buying? Summary of medical insurance prices, foreign generic drugs and efficacy
2024 In 2024, a piece of news about “the inclusion of new drugs for rare tumors in children into medical insurance” attracted great attention from the medical community and patients’ families. This drug is the MEK inhibitor selumetinib developed by AstraZeneca. It provides a new treatment method for children with symptoms related to neurofibromatosis type 1 (NF1).
As an important breakthrough in the field of targeted treatment of rare diseases in recent years, selumetinib has not only been approved for marketing internationally, but has also been successfully approved in China and included in the medical insurance directory, which has greatly alleviated the financial pressure on patients' families. So, what is the price of selumetinib? Is the efficacy trustworthy? Are there more affordable generic drugs? This article will provide an in-depth analysis around the hot issues that these patients are most concerned about.

1. Selumetinib: from breakthrough therapy to China’s medical insurance list
Selumetinib (Selumetinib) is an oral small molecule MEK inhibitor, first developed by AstraZeneca (AstraZeneca) and Merck Developed in collaboration with Merck (Merck), it is initially used to treat cancers related to RAS or BRAF mutations. In 2020, the U.S.FDA approved it for the treatment of 3 patients with inoperable plexiform neurofibromas (
The National Medical Products Administration of China (NMPA) accelerated the approval of selumetinib in 2023, approved its marketing in China, and was subsequently officially included in the medical insurance directory. This initiative allows domestic patients to use original drugs within an affordable range, truly reflecting the original intention of the "patient-centered" policy.
The launch of selumetinib not only solves the treatment gap, but also promotes the diagnosis and management of NF1 diseases in China into a new era.
2. Price Analysis: Comparison of Domestic Medical Insurance Prices and Laos Generic Drug Prices
At present, selumetinib can be prescribed in regular hospitals in China, and the patient's out-of-pocket payment is significantly reduced after medical insurance reimbursement. The original drug is priced at about more than 20,000 yuan per box, which fluctuates slightly depending on the hospital, region, and medical insurance policies. Calculated based on the medical insurance reimbursement ratio in some cities, the actual average monthly out-of-pocket expense for patients is about 10,000 yuan.
For patients without health insurance or who need long-term medication, this is still a significant expense. Therefore, some patients have begun to pay attention to the overseas generic drug market.
The price of the Laos generic version of selumetinib is much lower than that of the domestic original drug, about 2000 RMB. The active ingredients of the generic version are basically the same as those of the original drug, and some feedback has been accumulated in clinical use. Although generic drugs have not been approved by China's NMPA, some professional cross-border drug service platforms provide genuine channels for families with financial difficulties to consider using them under the guidance of doctors.
Of course, whether to choose generic drugs still requires patients to weigh comprehensively based on their actual condition, doctor’s advice, and financial status.
3. Efficacy evaluation: Clinical trials have proven that selumetinib can significantly shrink tumors
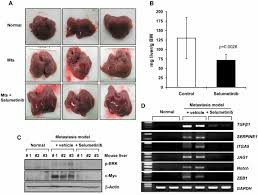
Effectiveness is an important basis for deciding whether patients should take medicine for a long time. According to the results of the SPRINT clinical trial published in the New England Journal of Medicine in 2020, selumetinib showed excellent results in the treatment of NF1-related plexiform neurofibromas:
More than 68% of children who received treatment had their tumors reduced in size by at least 20%;
Most children's symptoms improved significantly, such as pain relief, improved exercise ability, and improved quality of life;
Some children may even avoid surgery due to tumor compression.
It is worth noting that these effects mostly appear after several months of continuous medication, indicating that selumetinib is a long-term maintenance targeted therapy that requires regular medication and persistence in treatment.
In addition, selumetinib is also being studied for other diseases related to RAS pathway abnormalities (such as Noonan syndrome, low-grade glioma, etc.), indicating its potential for future expansion of indications.
4. Adverse reactions and safe use: controllable and mild
Selumetinib demonstrated a good safety profile in clinical trials. Common adverse reactions include:
1.Diarrhea, rash, fatigue, vomiting, oral ulcers, etc.;
2.A very small number of patients experience a slight decrease in cardiac function and an increase in liver enzymes;
3.Pediatric patients may be at risk of slowed growth and require regular monitoring of height and weight.
It is worth emphasizing that most of the side effects of selumetinib are reversible and can be alleviated by reducing the dose or briefly discontinuing the drug under the guidance of a doctor. Compared with the side effects of traditional chemotherapy drugs such as bone marrow suppression and hair loss, selumetinib has a higher overall tolerance and is particularly suitable for long-term use.
Clinicians also recommend the following monitoring during use:
1.Test liver and kidney function every1~2 months;
2.Carry out cardiac ultrasound every 3 months;
3.Regularly evaluate tumor volume and functional improvement.

5. True feedback from patients: The tumor has become smaller and the child no longer cries all night
According to feedback from many family members, the most intuitive change in children after using selumetinib is"No more crying all night." Many children suffer from persistent pain and functional disability due to tumor compression. After receiving selumetinib treatment, their pain is significantly reduced and their living conditions are significantly improved.
"My legs hurt when I took just two steps, but now I can go down the stairs by myself.", "My child's tumor shrank by one-third in three months, and the doctor said there is no need for surgery." Feedback like this is common among NF1 family members.
Of course, some patients experience mild gastrointestinal discomfort in the early stages of taking the medication, but the condition is relieved after adjusting their diet and using the medication.
Real feedback shows that although selumetinib cannot cure the disease, it has clear effects in delaying the disease, controlling tumor growth, and improving quality of life.
6. Suggestions and usage procedures for purchasing drugs at home and abroad
At present, domestic patients mainly have the following three channels to obtain selumetinib:
Purchasing prescriptions from medical insurance hospitals: Suitable for patients who have been diagnosed with NF1 and are included in medical insurance. They can enjoy medical insurance reimbursement and the financial burden is relatively light.
Purchasing original drugs at your own expense: suitable for patients who urgently need drugs but have not yet been reimbursed, or whose medical insurance reimbursement ratio is insufficient.
Cross-border drug purchase (Laos generic drugs): It is suitable for patients who are under great financial pressure and are willing to try generic drugs, but it should be done through formal institutions and close communication with the attending doctor should be maintained.
No matter which method you choose, it is recommended that all patients have medication evaluation and follow-up by a professional doctor. It is not recommended to buy or blindly try alternative medicines on your own.
The advent of selumetinib brings hope to children with NF1 related neurofibromas who have long lacked effective treatments. In terms of efficacy, it has been recognized as having the ability to significantly shrink tumors and improve the quality of life; in terms of price, it has been included in medical insurance in China, and there are also generic drugs abroad that provide a variety of choices, further improving drug accessibility.
In the context of an era in which "rare diseases are treatable and manageable" are gradually being realized, the success of selumetinib represents an important shift in innovative drugs from "out of reach" to "accessible and affordable".
ForNF1For patient families, scientific selection of medication routes, standardized medication, and regular follow-up are key steps to achieve the best treatment effect. As more real-world data accumulates, selumetinib deserves continued attention in terms of indication expansion and treatment optimization in the future.
xa0
References:
1.The New England Journal of Medicine – “Selumetinib in Children with Inoperable Plexiform Neurofibromas”
2.FDADrug Approval Information – FDA.gov
3.National Medical Insurance Administration Drug Catalog (2023 edition) – nhsa.gov.cn
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















