What is the availability and market price of mitomycin intravesical solution (Zusduri) in China?
Mitomycin Intravesical Solution (ZUSDURI) byUroGen Developed by PharmaCompany, it is the world's first local chemotherapy drug using RTGel® delivery technology. It is specially designed to treat low-grade, recurrent intermediate-risk non-muscle-invasive bladder cancer (LG-IR-NMIBC). In June 2025 June , the US FDA has officially The drug is officially approved for use in adult patients who do not require cystectomy. It provides a new alternative to transurethral tumor resection (TURBT) and marks a new stage in the local treatment of bladder cancer.
Up to now, mitomycin intravesical solution (ZUSDURI) has not been registered with the China National Food and Drug Administration (NMPA), nor has it been included in the Chinese Clinical Trial Publicity Catalog. This means that the drug has not yet obtained the qualification for legal sale and use in China. At present, patients with bladder cancer in China still rely on traditional transurethral surgery, intravesical BCG instillation or conventional chemotherapy, and there is no clinically accessible channel for ZUSDURI.
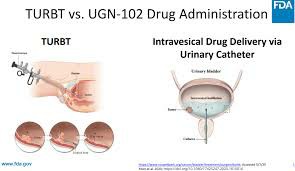
Although ZUSDURI plans to be officially launched in the United States in 2025July, the official price has not been announced so far. However, because it is an innovative delivery platform, does not require surgery, and can be operated in an outpatient setting, it is generally expected that the price of a single course of treatment will be high, possibly reaching the level of thousands to tens of thousands of dollars. Future prices will be affected by medical insurance reimbursement policies and hospital procurement systems.
SinceZUSDURI is not yet available in China, patients cannot obtain the drug through hospitals or pharmacies. If there is a need for treatment, it can only be purchased through overseas channels, but it may face high costs and use risks. If the drug is applied for marketing in the country in the future, whether it will enter the medical insurance catalog, pricing mechanism and local access policy will directly determine the accessibility and economic burden of Chinese patients, which deserves great attention.
Reference materials:https://www.fda.gov/drugs/
xa0
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















