Summary of the scope and indications of mitomycin intravesical solution (Zusduri) for the treatment of bladder cancer
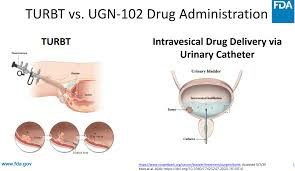
Mitomycin intravesical solution (Zusduri) is a drug designed to treat non-muscle invasive bladder cancer (NMIBC), especially in low-grade, recurrent, intermediate-risk patient groups. Although the tumors in these patients have not invaded the bladder muscle layer, they have a tendency to recur repeatedly. Traditionally, they need to undergo multiple transurethral resections of bladder tumors (TURBT), which has limited efficacy and a high recurrence rate. The launch of Zusduri provides these patients with a new option for non-surgical, local treatment.
It should be noted that Zusduri is currently only suitable for low-grade, intermediate-risk tumors that do not involve the bladder muscle layer, and is not currently suitable for high-grade, muscle-invasive (MIBC) or metastatic bladder cancer cases. For such advanced patients, systemic chemotherapy, radiotherapy or total cystectomy are still the main treatment strategies. Therefore, the use of Zusduri should be strictly screened and evaluated by professional urologists based on tumor stage and patient status.
Reference: https://www.fda.gov/drugs/
Zusduri's core target audience is patients who are not suitable for cystectomy or repeated TURBT surgery due to age, physical condition or other diseases. It is administered through bladder instillation once a week for 6 consecutive weeks to reduce the impact on the whole body and is especially suitable for people at high risk of surgery. Research shows that its complete remission rate is as high as 78%, and it has good ability to maintain remission and significantly delay the time of recurrence.
It should be noted that Zusduri is currently only suitable for low-grade, intermediate-risk tumors that do not involve the bladder muscle layer, and is not currently suitable for high-grade, muscle-invasive (MIBC) or metastatic bladder cancer cases. For such advanced patients, systemic chemotherapy, radiotherapy or total cystectomy are still the main treatment strategies. Therefore, the use of Zusduri should be strictly screened and evaluated by professional urologists based on tumor stage and patient status.
Reference: https://www.fda.gov/drugs/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















