How effective is mitomycin intravesical solution (Zusduri) in the treatment of bladder cancer?
Mitomycin intravesical solution (Zusduri) is the world's first approved thermosensitive gel-delivered local chemotherapy drug, specifically designed to treat non-muscle invasive bladder cancer (NMIBC). Compared with traditional repeated surgery (TURBT), Zusduri provides a "non-invasive" medical treatment option, especially suitable for low-grade, intermediate-risk patients with recurrence. Its listing breaks the long-term pattern of NMIBC treatment relying on surgery and BCG infusion, providing new hope for patients who are not suitable or unwilling to undergo surgery.
Based on data from the pivotal ATLAS and ENVISION clinical studies, Zusduri demonstrated significant efficacy in the treatment of relapsed LG-IR-NMIBC patients. At the end of 6 a standard course of infusion therapy, approximately 78% of patients achieved cystoscopic complete remission (
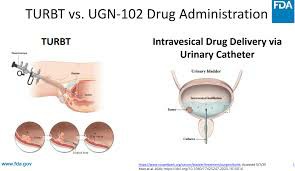
The efficacy of Zusduri provides clinical evidence support for "drug replacement surgery". Compared with the bladder wall damage and complications caused by repeated administration of TURBT, Zusduri uses local sustained-release technology to increase the residence time of the drug in the bladder, effectively killing cancer cells and reducing the recurrence rate. Zusduri is becoming a practical treatment option for patients who cannot tolerate multiple surgeries or who wish to preserve bladder function.
AlthoughZusduri currently performs well in terms of efficacy, its long-term use data is still being accumulated, especially its adaptability to different stages and pathological types still needs further verification. In addition, this drug has not yet been included in the medical insurance catalogs of most countries, and the cost of use is high, which may affect its widespread popularity to a certain extent. However, as more research results are released and medical insurance negotiations gradually advance, Zusduri is expected to become NMIBCOne of the important pillars of management.
Reference materials:https://www.fda.gov/drugs/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















