"Precision targeting + combination therapy" sets off a new trend: Ivosidenib may bring hope to patients with AML and cholangiocarcinoma?
2025June In June, new news came from the American Society of Clinical Oncology (ASCO) annual meeting. A new study for acute myeloid leukemia (
So why has Avonib attracted widespread attention around the world? What is its scope of indications, treatment mechanism, progress in combination therapy, efficacy data and patient costs? This article will systematically interpret this "star targeted drug" and provide detailed reference for domestic patients.
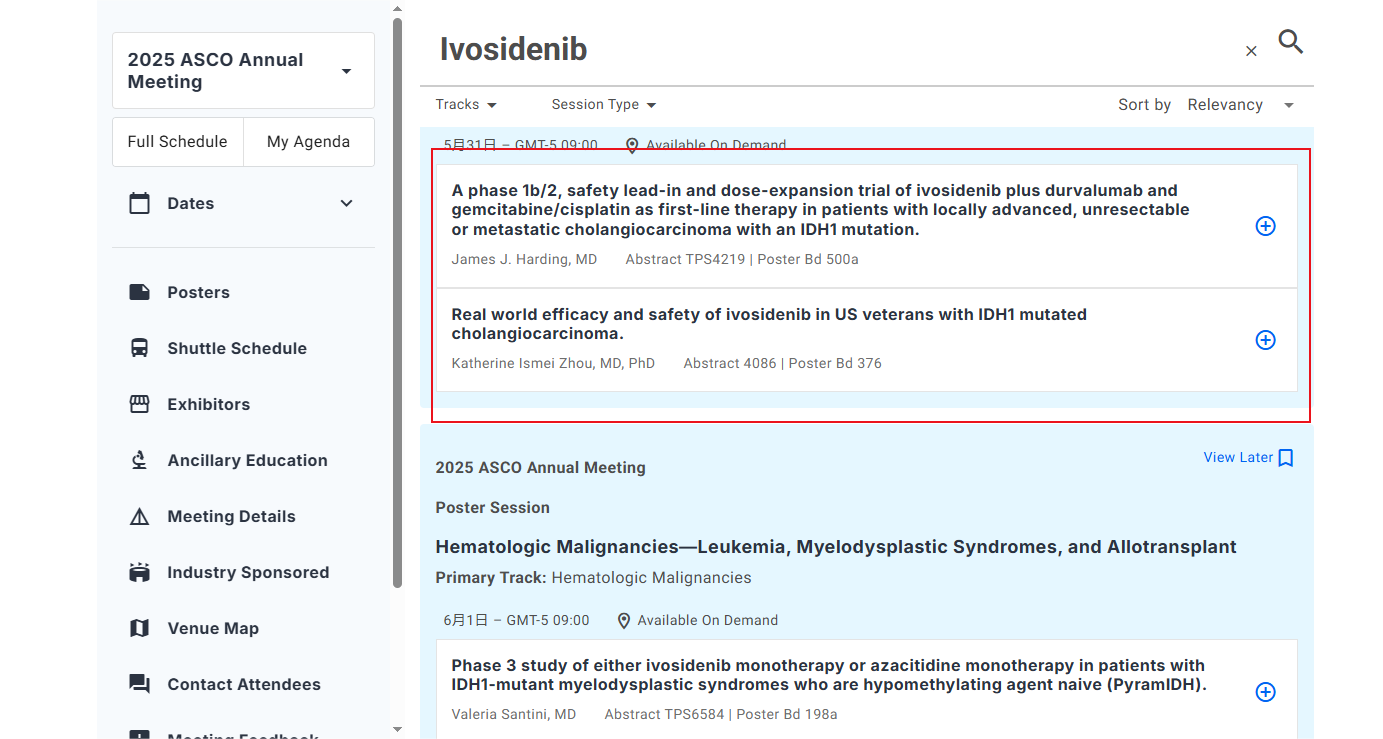
ASCO Focus again at the annual meeting: ivonib combined with azacitidine significantly prolongs AML survival
At the2025ASCO annual meeting, researchers announced the AGILE 3 year follow-up data. This study compared the efficacy of ivosidenib + azacitidine (Ivosidenib+Azacitidine) with azacitidine alone in treatment-naïve older patients with AML who carry the IDH1 mutation. The data shows that the median overall survival time of the combination group reached 29.3 months, while that of the single-drug group was 7.9 months. The survival period was extended by 21.4 months!
In addition, the complete response rate (CR) was also significantly improved, reaching 47% in the combination group, but only in the azacitidine group15%. The researchers pointed out that ivonib inhibits the activity of IDH1 mutant enzyme, reduces the tumor metabolite 2-HG, and restores hematopoietic differentiation. It is a precise and well-tolerated targeted solution.
The results of this study have triggered strong responses from the clinical community and have been included in recommended standards in guidelines from many countries. They are especially suitable for elderly or frail patients who cannot tolerate intensive chemotherapyAML.
IDH1Mutation: The culprit hidden behind cancer, targeted therapy emerges in response
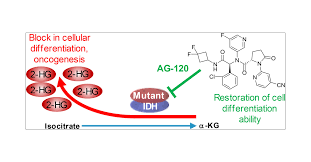
IDH1 (isocitrate dehydrogenase1) is an enzyme involved in cell metabolism. When this gene is mutated, it will lead to intracellular metabolic disorders and abnormality. The metabolite 2-hydroxyglutarate (2-HG) affects the normal differentiation of hematopoietic stem cells and induces leukemia or solid tumors.
According to statistics, about6%-10% ofAML patients have IDH1 mutations, and in cholangiocarcinoma, glioma, and soft tissue sarcoma, the detection rate of IDH1 mutations also reaches 10%-30%. Because these cancer types carry specific mutations, traditional chemotherapy is often ineffective. The emergence of IDH1 inhibitors such as ivonib is filling this treatment gap.
In 2018, the United StatesFDA approved the marketing of ivonib for the treatment of relapsed / refractory AML carrying IDH1 mutations. In 2021, FDA further approved its combination with azacitidine for initial treatment of IDH1 mutated AML patients who are not suitable for intensive chemotherapy. 2023In 2017, ivonib was granted breakthrough therapy status to explore the treatment of IDH1-mutated cholangiocarcinoma, and its clinical application landscape continued to expand.
Multiple indications have made breakthroughs: AML, cholangiocarcinoma, and glioma have promising prospects
1. Acute myeloid leukemia (AML)
In addition to the AGILE study, the IDHENTIFY study also showed that the recurrence of IDH1 mutations< /span>/RefractoryAML patients can achieve a complete remission rate of 24.7%. Some patients do not need hematopoietic stem cell transplantation and directly achieve lasting remission after treatment.
In addition, the "low-toxic triple regimen" composed of ivonib, venetoclax (Venetoclax) and low-dose cytarabine (LDAC) has also shown good safety and efficacy in multiple exploratory studies, and is expected to become a new regimen more widely applicable to the elderly.
2. Cholangiocarcinoma (Cholangiocarcinoma)
2021ClarIDHy trial announced, median progression-free survival of patients with IDH1 mutated cholangiocarcinoma treated with ivonib The median overall survival period (PFS) was 2.7 months, and that of the control group was 1.4 months. The median overall survival was also significantly prolonged. This result prompted the FDA to grant ivonib "breakthrough therapy designation" and be included in the NCCN guidelines as a recommended drug, especially for patients who have failed second-line treatment.
3. Glioma (Glioma)
IDH1mutated low-grade glioma (LGG) is a relatively slow-progressing but difficult-to-treat disease. Early clinical data shows that ivonib can delay tumor progression and has mild side effects. In the future, it may be further expanded to an earlier intervention stage to improve patients' quality of life.
A concise explanation of the medication mechanism: Precisely blocking the carcinogenic pathway of 2-HG
Avonib is an oral small molecule IDH1 inhibitor that can specifically bind to the mutant IDH1 protein and block its ability to produce the carcinogenic metabolite 2-HG. The accumulation of 2-HG can inhibit the differentiation of hematopoietic cells and even trigger the "reversion" of tumor cells.
By reducing 2-HG concentrations, ivonib can restore hematopoietic differentiation, reduce tumor burden, and induce remission. Its precise mechanism gives it the following advantages:
1.The function is clear and the target is clear;
2.Low toxic and side effects, suitable for elderly or frail people;
3.Can be used alone or in combination, with high flexibility;
4. There is no need for hospitalization and it can be taken orally to improve patient compliance.
Side effects and medication precautions
According to clinical trials and real-world data, ivonib is generally well tolerated, and common adverse reactions include:
1.Leukopenia and anemia;
2.Nausea, diarrhea, constipation;
3.Elevated creatinine and elevated liver enzymes;
4.Differentiation Syndrome: occurs in about 10%-15% of patients and can be alleviated by glucocorticoid intervention.
In addition, electrolytes, liver and kidney function, and bone marrow status need to be monitored regularly during ivonib treatment to determine efficacy and adjust medication.
Avosidenib price and acquisition method: original research is the main focus, and generic drugs are more suitable for economical ordinary patients!
Currently, ivonib is an original drug developed by Agios Pharmaceuticals and is commercialized and promoted by Servier. IvonibThe original drug is currently on the market in China, but it is not included in medical insurance. The price is as high as about 30,000 to 40,000 yuan. Please consult the local hospital pharmacy for specific prices.
Overseas price reference:
1.US price: about$26,000/month (250mgonce a day, each bottle60tablets)
2.Laos Generic version: Laos Lucius version generic drug The price is about3000-4000RMB/month
If domestic patients consider using it, they must obtain a diagnosis certificate of indications and a genetic test report (IDH1 mutation positive) from a doctor, and purchase the drug through formal channels.
In the era of precision, ivonib will become the "backbone" in the treatment of IDH1 mutated tumors
FromAMLFrom its significant survival benefit to its breakthrough efficacy in cholangiocarcinoma to its potential value in glioma, ivonib has gradually established a new treatment pattern of "IDH1mutation+ targeted intervention". As precision medicine becomes increasingly mature, it may become an indispensable targeting option for specific groups of people.
For patients, receiving genetic testing and clarifyingIDH1 mutation status is the first step into a new era of targeted therapy. In the future, with the launch of more generic drugs and the advancement of medical insurance policies, ivonib is expected to benefit more domestic patients.
xa0
References:
1.ASCO 2025Annual Meeting Research Summary:https://meetings.asco.org
2.ClarIDHy Trial - N Engl J Med 2020; 382:817-828:https://www.nejm.org/doi/full/10.1056/NEJMoa1913006
3.AGILE Trial - Lancet Oncol 2022; 23: 676–88:https://doi.org/10.1016/S1470-2045(22)00127-X
4.FDA Drug Approval Summary - Ivosidenib:https://www.accessdata.fda.gov
5.NCCN Guidelines - AML, Cholangiocarcinoma (IDH1 Mutations) 2024:https://www.nccn.org
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















