How to buy EIKANCE 0.01% Atropine Sulfate Eye Drops through regular channels and precautions
As of2025, 0.01% atropine sulfate eye drops (EIKANCE) has not been officially launched in China, and it has not been included in the medical insurance catalog. Therefore, the drug cannot be purchased directly in domestic hospitals or pharmacies. For children with myopia who need medicine, their families usually need to make cross-border purchases through regular overseas pharmacies or third-party drug service platforms, provided they have a valid prescription issued by a doctor. It is recommended to communicate with a professional doctor before purchasing to confirm the necessity and appropriate dosage.
EIKANCE is a prescription drug product developed by an Australian pharmaceutical company. It has been approved by the Australian TGA and can be purchased in Australian pharmacies with a prescription. Common overseas specifications are 0.01% concentration, and each box contains 30 0.3ml small ampoules. The price may be around 600 yuan (the price will be affected by exchange rate fluctuations). In addition to the Australian version, there is also a generic version in India. The specification is 0.01% w/v*5ml bottles. The price is relatively low, about tens of yuan per bottle. Regardless of whether you choose original research or generic drugs, it is recommended to import them through formal channels and pay attention to check the authenticity of the drugs.
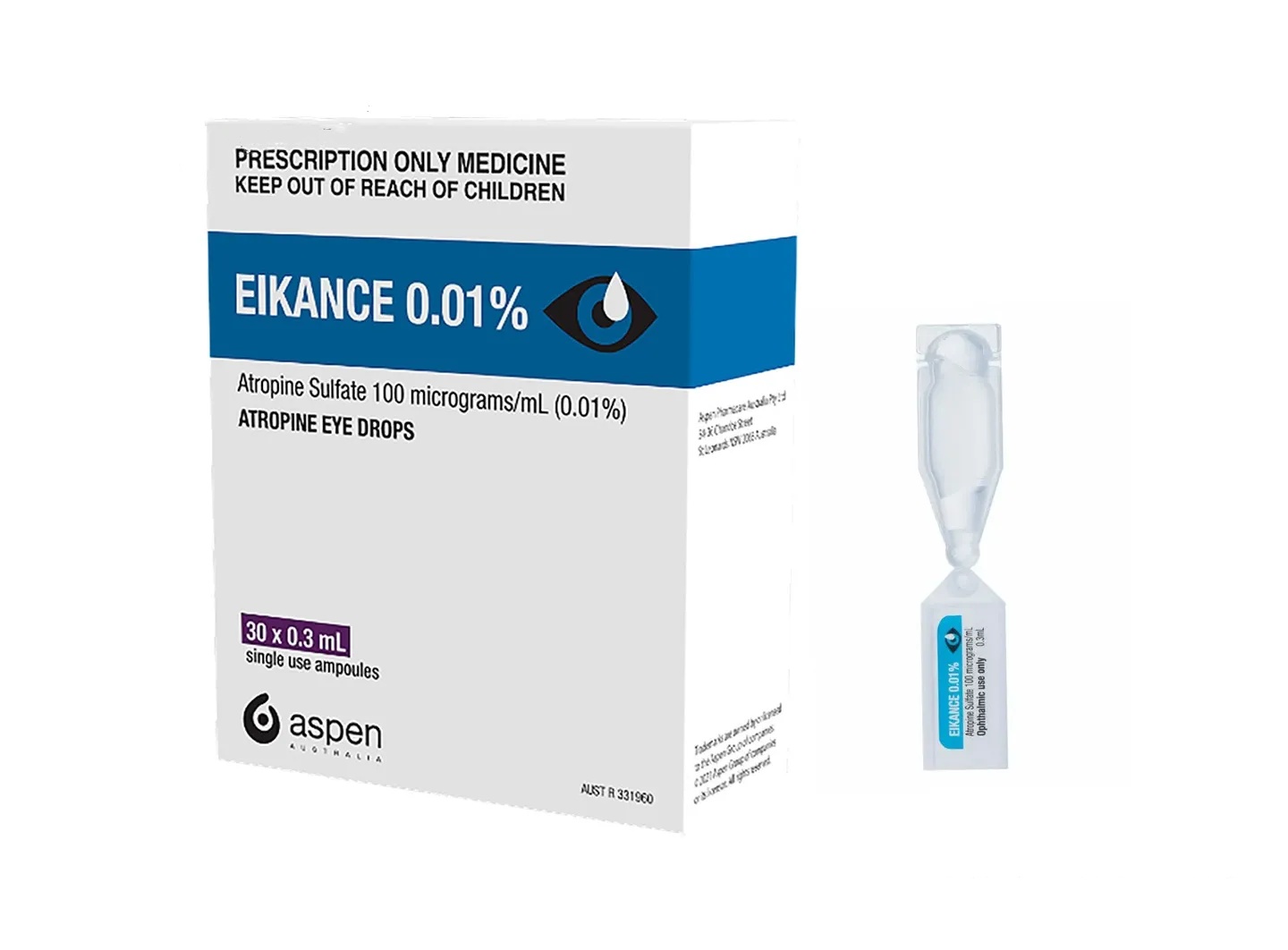
EIKANCE are low-concentration atropine sulfate eye drops that need to be used once a day, usually one drop in each eye before going to bed. Drugs should be stored in a cool and dark place away from light. Some versions recommend refrigeration (2-8°C) to extend stability. Each bottle is for one-time use and cannot be reused after opening. During use, care should be taken to avoid contaminating the dripper, and eyes should be closed for 1-2 minutes after dripping to enhance absorption. If discomfort such as dry eyes, photophobia or allergies occurs, stop using it immediately and consult a doctor.
SinceEIKANCE has not yet been registered in China, parents should ensure that the source of drugs is formal when choosing overseas channels to purchase, and keep credentials such as prescriptions, drug lists, and transportation records. Do not obtain medicines through unknown platforms or personal purchasing channels to avoid buying counterfeit or improperly stored products. During the medication process, changes in eye axis and vision should be reviewed regularly, the treatment effect should be evaluated, and the plan should be adjusted in a timely manner to ensure safe and effective prevention and control of myopia in children.
Reference materials:https://www.medsafe.govt.nz/profs/datasheet/e/EikanceEyeDrops.pdf
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















