Avmapki and Faczynja combination description application guide and market status
1. Name:avutometinib and defactinib (capsules/tablets)
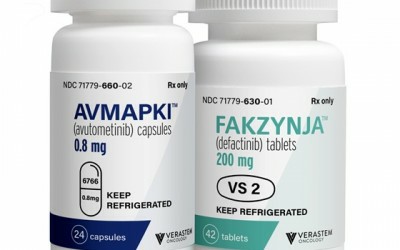
Product name:Avmapki and Faczynja combination
2. Indications:
The combination of Avmapki and Faczynja is indicated for the treatment of adult patients with KRAS-mutated recurrent low-grade serous ovarian cancer (LGSOC) who have received prior systemic therapy.
3. Usage and dosage:
1. Patient selection: Patients with recurrent LGSOC treated with Avmapki-Faczynja combination are selected based on the presence of KRAS mutations in tumor specimens. There are currently no FDA-approved tests to detect KRAS mutations in LGSOC for use in selecting patients to receive the Avmapki-Faczynja combination.
2. Recommended dosage and administration
(1) AVMAPKI capsules: The recommended dose of capsules is 3.2 mg (four 0.8 mg capsules) taken orally twice weekly (days 1 and 4) for the first 3 weeks of each 4-week cycle until disease progression or unacceptable toxicity. Take AVMPKI with each dose and should be taken with food. Swallow capsule whole. Do not chew, break or open capsules.
If a dose is missed by more than 24 hours, skip the missed dose and take the next scheduled dose as prescribed. Do not take both doses at the same time to make up for a missed dose. If you experience vomiting after taking AVMAPKI, do not take additional doses. Take your next scheduled dose as prescribed.
(2) FAKZYNJA Tablets: The recommended dose of tablets is 200 mg (one tablet) taken orally twice daily for the first 3 weeks of each 4-week cycle until disease progression or unacceptable toxicity. Take the tablets with food and swallow the tablets whole. Do not chew, break, or crush tablets.
If a dose is missed by more than 6 hours, skip the missed dose and take the next scheduled dose as prescribed. Do not take both tablets at the same time to make up for a missed dose. If vomiting occurs after taking this dose, do not take additional doses. Take your next scheduled dose as prescribed.
3. Eye examination and preventive skin medications
(1) Eye examination: Regardless of baseline examination results, perform a comprehensive eye examination before Cycle 2 and every three cycles thereafter and as clinically indicated.
(2) Medications to prevent skin reactions: at the beginning and during at least the first 2 cycles of using Avmapki-Faczynja combination:
•Topical corticosteroids (applied to face, scalp, neck, upper chest and upper back)
•Systemic oral antibiotics
4. Dose adjustment: Due to possible adverse reactions caused by the use ofAvmapki-Faczynja combination, its dose may need to be adjusted under the guidance of a doctor. The dose of AVMAPKI capsules can be reduced to 2.4 mg twice weekly for the first 3 weeks of every 4-week cycle; the dose of FAKZYNJA tablets can be reduced to 200 mg orally once daily for the first 3 weeks of every 4-week cycle; for patients who are still intolerant after one dose reduction, permanently discontinue the Avmapki-Faczynja combination.
4. Adverse reactions:
In clinical studies ofAvmapki-Faczynja combination, common adverse reactions included nausea, fatigue, rash, diarrhea, musculoskeletal pain, edema, vomiting, abdominal pain, dyspepsia, acneiform dermatitis, vitreoretinal disease, stomatitis, pruritus, visual impairment, constipation, dry skin, dyspnea, cough, and urinary tract infection. Common laboratory abnormalities include increased creatine phosphokinase, increased aspartate aminotransferase, decreased hemoglobin, increased alanine aminotransferase, increased blood bilirubin, increased triglycerides, decreased lymphocyte count, increased alkaline phosphatase, decreased platelet count, and decreased neutrophil count.
5. Supply and storage:
AVMAPKI is available as 0.8 mg capsules and Faczynja as 200 mg tablets. Keep AVMAPKI capsules and FAKZYNJA tablets in the refrigerator at 2°C to 8°C (36°F to 46°F).
6. Mechanism of action:
Avutometinib is a MEK1 inhibitor. Avutometinib induces the formation of inactive RAF/MEK complexes and prevents phosphorylation of MEK1/2 by RAF. RAF and MEK proteins are regulators of the RAS/RAF/MEK/ERK (MAPK) pathway. Avutometinib inhibits MEK1/2 and ERK1/2 phosphorylation and proliferation of tumor cell lines harboring KRAS mutations. Treatment of cancer cells with Avutometinib increases levels of phosphorylated focal adhesion kinase (FAK).
Defactinib is an inhibitor of FAK and proline-rich tyrosine kinase-2 (Pyk2), two non-receptor tyrosine kinases in the FAK family. Defactinib inhibits FAK autophosphorylation in cancer cells in vitro and in mouse xenograft models.
Avutometinib combined with Defactinib enhances in vitro cell proliferation inhibition and anti-tumor activity in mouse tumor models, including LGSOC.
7. Listing situation:
On May 8, 2025, the Avmapki-Faczynja combination was approved by the U.S. Food and Drug Administration (FDA) and will be sold in the United States.
Reference materials:https://www.verastem.com/pdf/avmapki-faczynja-co-pack-full-prescribing-information.pdf
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















