On May 9, 2025, Johnson & Johnson released the latest research results of its oral peptide antagonist Icotrokinra (codename JNJ-2113) in the phase III clinical trial ICONIC-TOTAL. The data showed that Icotrokinra showed significant advantages in improving skin clearance in patients with moderate plaque psoriasis, especially when treating difficult-to-treat areas such as the scalp and genitals, achieving both primary and secondary efficacy endpoints. These results will be highlighted at the 2025 Society for Investigation of Dermatology (SID) Annual Meeting in San Diego, California. Plaque psoriasis is an immune-mediated chronic inflammatory disease characterized by abnormal proliferation of skin cells, leading to the formation of erythema and scales, often accompanied by itching and pain. The scalp and genitals are common and difficult-to-treat areas of psoriasis, with high numbers of patients affected and traditional treatments such as topical medications and phototherapy having limited efficacy in these areas.
Icotrokinra is an innovative, selective IL-23 receptor (IL-23R) antagonist in the form of an oral peptide drug. It can specifically bind IL-23R with extremely high affinity and significantly inhibit the activity of IL-23 signaling pathway in human T cells. Compared with existing biological agents or non-specific systemic treatments, Icotrokinra brings a new oral treatment option to patients, which not only ensures systemic efficacy, but also enables the convenience of once-daily administration. The drug's unique mechanism offers new therapeutic hope for psoriasis patients, especially those who have not responded well to traditional treatments.
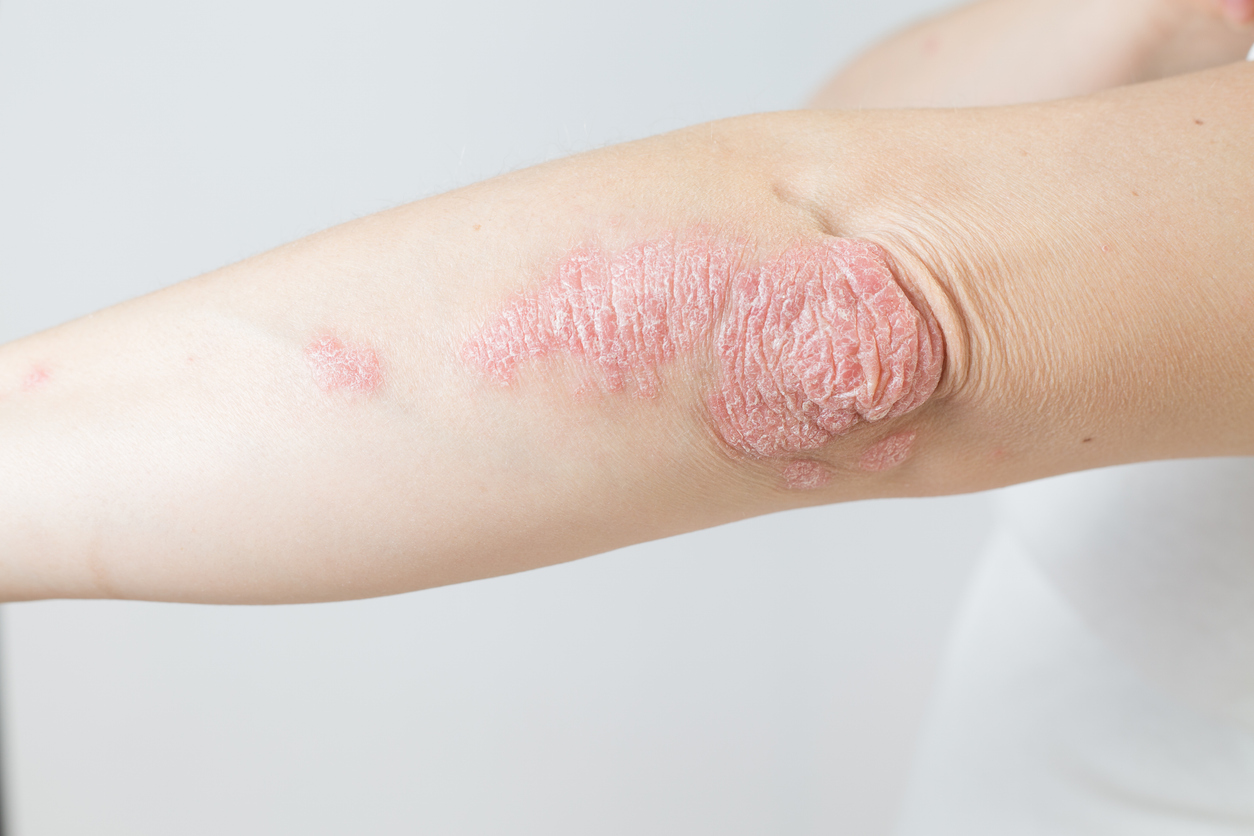
The ICONIC-TOTAL trial is a randomized, controlled, phase III clinical trial involving 311 patients with moderate or above plaque psoriasis, all of whom had special affected areas such as the scalp, genitals, hands and feet. Of the subjects, 208 received Icotrokinra and 103 received a placebo. The primary endpoint of the trial was a grade 2 or above improvement in clinical global assessment (IGA) at 16 weeks. The results showed that 57% of the patients in the Icotrokinra group achieved the primary endpoint, while only 6% in the placebo group, the difference was extremely significant (P<0.001). In addition, among patients with scalp psoriasis, 66% of the Icotrokinra treatment group achieved a scalp-specific assessment of 0/1 (skin is clear or almost clear), far exceeding the 11% of placebo; among patients with genital psoriasis, 77% of Icotrokinra patients achieved a static physician assessment of 0/1, significantly higher than the 21% of placebo. In the subgroup with hand and foot involvement, 42% of patients achieved clarity or almost clarity, compared with only 26% in the placebo group. These data fully demonstrate the broad efficacy of Icotrokinra in multi-site psoriasis.
In terms of safety, Icotrokinra was well tolerated. During the 16 weeks of the trial, the incidence rates of adverse events in the Icotrokinra group and the placebo group were 50% and 42% respectively, and the proportions of serious adverse events were 0.5% and 1.9% respectively. There were no new safety signals. Johnson & Johnson stated that Icotrokinra is not only in the critical Phase III clinical development stage in moderate to severe plaque psoriasis, but is also in phase 2b trials in the research of active psoriatic arthritis and moderate to severe active ulcerative colitis, and it is expected to have broad application potential in the treatment of immune-related diseases.
Reference: Icotrokinra results show significant skin clearance in patients with difficult-to-treat scalp and genital psoriasis. News release. PR Newswire. Published May 9, 2025. Accessed May 9, 2025.

.jpeg)


















