Detailed explanation and application guide of Apelvis-Piqray’s medical insurance reimbursement policy
Alpelisib(Alpelisib)-Piqray is a new targeted drug used to treat HR-positive, HER2-negative advanced or metastatic breast cancer. However, the original drug of Apelvis is not currently on the market in China, and therefore has not yet been included in the national medical insurance system. This means that patients may face a higher financial burden when obtaining treatment with these drugs.
Since Apelis has not yet entered the domestic market, the specific selling price is not yet clear. Usually, the price of drugs will vary according to factors such as manufacturer, market demand, and import-related policies. In addition, since Apelvis is a relatively new therapy, its price may receive greater attention after its launch, which also affects patients' purchasing decisions.
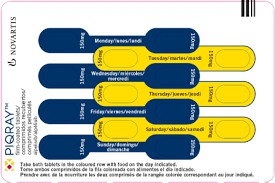
In terms of application guidelines, although Apelvis is not reimbursed by medical insurance, patients can still refer to its indications and usage when seeking treatment. Doctors will take into account the patient's specific condition and past treatment responses when formulating a treatment plan for the patient. Under the guidance of a doctor, patients should conduct regular follow-up visits to monitor efficacy and adverse reactions, and make dose adjustments as needed.
Patients choosing Apelvis as a treatment option should fully discuss the potential benefits and risks with their doctor, especially with regard to affordability. If the patient's financial resources are limited, the doctor may recommend exploring other treatment options or clinical trials to find more cost-effective alternative treatments.
In addition, with the increasing clinical application of Apelvis, it is expected that the accumulation of clinical data will promote its inclusion in medical insurance in the future. Patients and their families can actively pay attention to relevant policy developments and understand the latest adjustments and information on medical insurance, thereby providing a basis for subsequent treatment decisions.
Reference materials:https://www.piqray.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















