Mitotane is produced by which specific manufacturer?
As an important drug for the treatment of adrenocortical cancer (adrenal gland cancer), Mitotane has relatively limited manufacturers around the world. It is mainly manufactured and supplied by a few pharmaceutical companies that specialize in the development and production of anti-tumor drugs. One of the world's well-known mitotane manufacturers is the Greek pharmaceutical company HRA Pharma. The company has a high reputation in the field of anti-tumor drugs and its mitotane products are widely used in clinical applications, especially in Europe and some Asian markets. In addition, the production of mitotane also involves a few generic drug manufacturers in other regions, but these are mostly regional manufacturers with relatively limited production scale and market coverage.
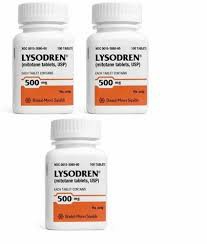
The production requirements of Mitotane are relatively high. Because of its special drug properties, it is a highly fat-soluble steroid compound. The stability and purity of the preparation directly affect the clinical effect and safety of the drug. Therefore, the world's major manufacturers of mitotane strictly followGMP (Good Manufacturing Practice) standards to ensure that drug quality meets international requirements. As the main original manufacturer of mitotane, HRA Pharma is famous for its high purity and stability. It is accompanied by a complete clinical support and drug management system to provide patients with professional medication guidance.
In the Chinese market, mitotane has not yet formed large-scale domestic production. Original research products mainly rely on imports, and the supply channels are mostly imported and distributed by authorized agents. Some domestic pharmaceutical companies are actively exploring the development of generic versions of mitotane, but there is currently no widely marketed domestic version. When patients purchase mitotane, they should obtain it through formal medical institutions or hospital pharmacies and avoid purchasing drugs through informal channels to ensure drug quality and medication safety.
Reference materials:https://go.drugbank.com/drugs/DB00648
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















