What is the latest market price of margetuximab in 2025?
Margetuximab (margetuximab-cmkb) is a targeted immunotherapy drug for HER2-positive solid tumors. It is mainly used to treat patients with HER2-positive advanced or metastatic breast cancer, especially those whose disease has progressed after receiving multiple anti-HER2 treatments. By binding to the HER2 receptor and enhancing antibody-dependent cell-mediated cytotoxicity (ADCC) through an engineered Fc fragment, it has greater efficacy than traditional trastuzumab in activating immune cells to eliminate tumor cells.
This mechanism makes margetuximab particularly suitable for dealing with trastuzumab resistance, further extending the progression-free survival and overall survival of patients. In addition to its application in the field of breast cancer, margituximab is also actively exploring its potential for use in other HER2 expression-related cancers such as gastric cancer. Multiple international clinical trials are ongoing, showing its broad therapeutic prospects.
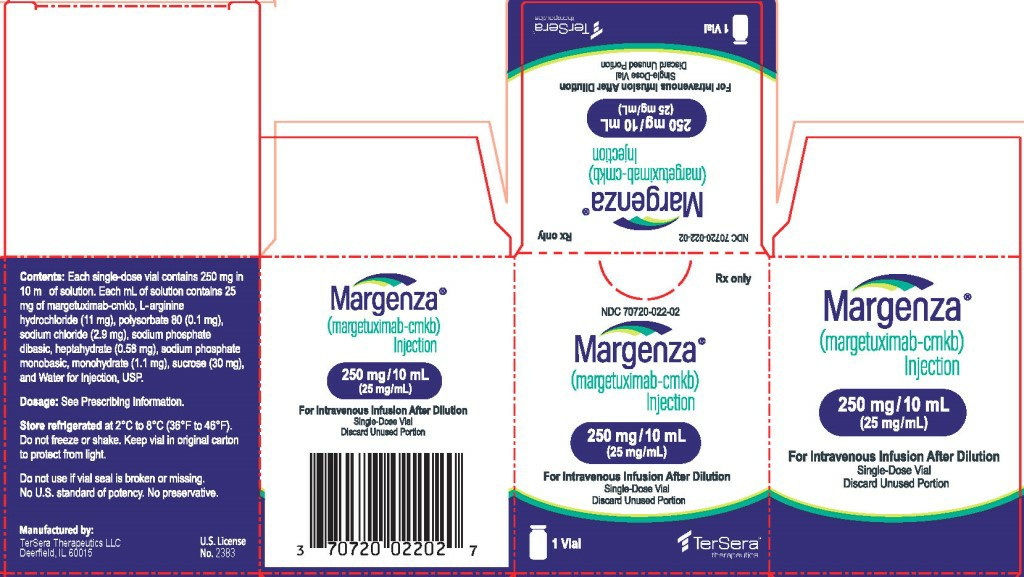
Regarding the market price, the original drug version of margetuximab in China is currently on the market. However, due to the short time to market, its specific market price has not yet been fully disclosed and transparent, and it has not yet been included in the national medical insurance directory. In the international market, magituximab has been approved by the European Medicines Agency (EMA) and other regions, providing stable supply. The common specifications overseas are 250mg/10ml bottle/box, and the price is about 30,000 yuan. However, the specific price will be affected by the exchange rate, taxes and distribution channels at the time, and may fluctuate to a certain extent. Currently, there are no generic drugs of margituximab on the domestic and foreign markets, and only the original version of the drug is available to patients. This also means that the quality and efficacy of the drug are highly guaranteed, but the cost burden is relatively high
Reference materials:https://www.margenza.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















