Voyxact (sibeprenlimab-szsi) instruction manual Chinese introduction
1. Common names: Voyxact, sibeprenlimab-szsi
2. Indications:
Voyxact (sibprenlimab-szsi) reduces proteinuria in adult patients with primary immunoglobulin A nephropathy (IgAN) who are at risk for disease progression. This indication received accelerated approval based on reduction in proteinuria. Whether VOYXACT can long-term slow the decline of renal function in patients with IgAN has not been determined. Continued approval for this indication may be contingent upon verification and characterization of clinical benefit in confirmatory clinical trials.
3. Usage and dosage:
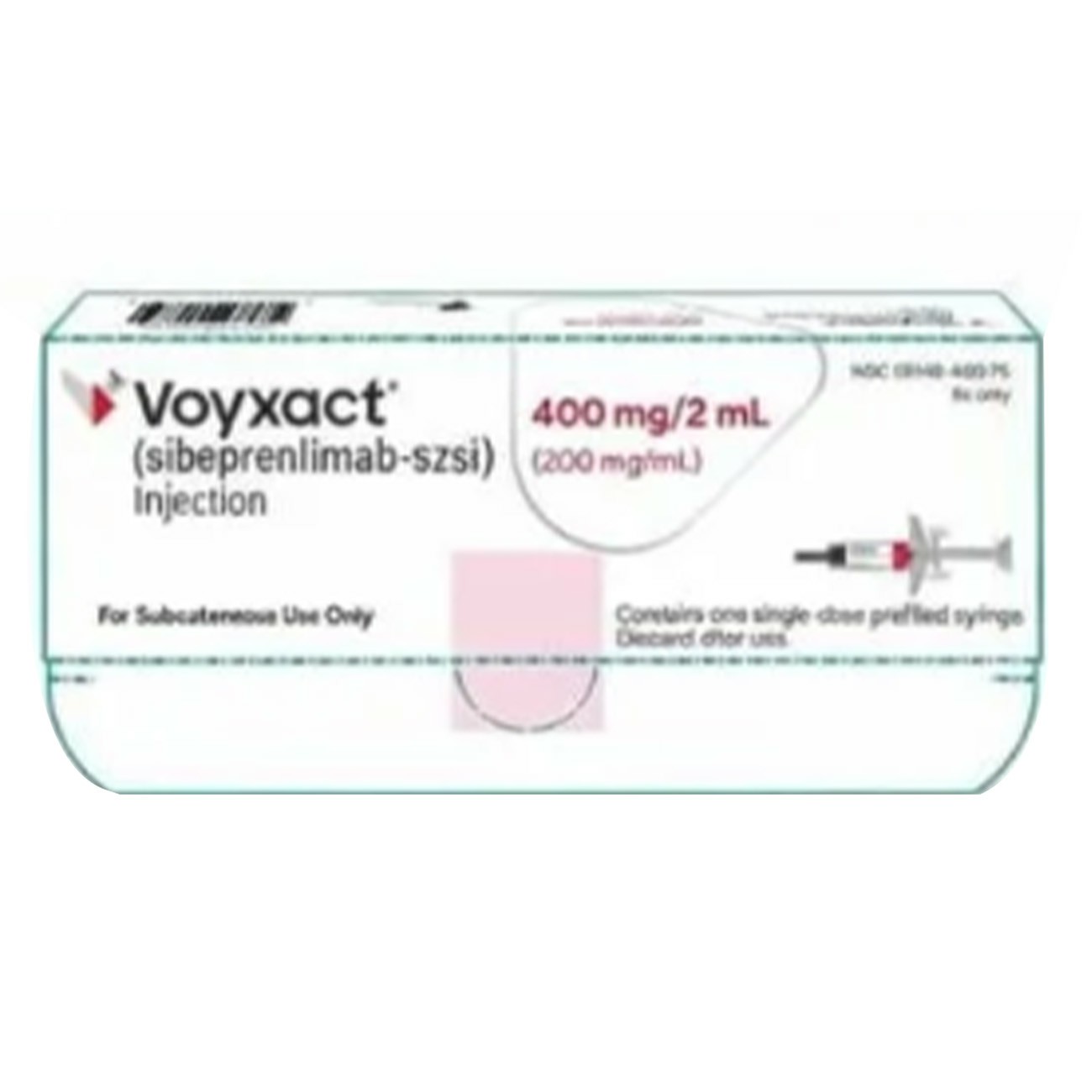
1. Recommended dose : The recommended dose of Voyxact is 400mg, injected subcutaneously every 4 weeks.
2. Missed dose: If you miss the scheduled dose of Voyxact, you should use the missed dose as soon as possible and then re-use it every 4 weeks.
3. Preparation and administration : Voyxact is for self-administration by patients or administration by caregivers. Prior to use, provide patients and/or caregivers with appropriate training regarding the use of Voyxact in accordance with the Instructions for Use. Visually inspect prefilled syringes for particulate matter and discoloration. The solution should be clear to milky white and colorless to yellow. Do not use prefilled syringes if the solution contains visible particulate matter, is turbid, or is discolored (except clear to opalescent, colorless to yellow).
Administered by subcutaneous injection only. Inject into the front of the thigh or abdomen. If a subcutaneous injection is given by a caregiver, the back of the upper arm can also be used as the injection site. Do not inject into the same site as a previous injection, or into a mole, scar, bruise, or area of u200bu200bskin that is tender, damaged, red, scaly, or hard.
4. Adverse reactions:
In clinical studies of Voyxact, common adverse reactions included upper respiratory tract infection and injection site erythema; warnings and precautions related to Voyxact include immunosuppression and increased risk of infection.
5. Supply and storage:
Voyxact injection is a sterile, preservative-free, clear to opalescent, colorless to yellow solution available in a single-dose prefilled syringe. Store Voyxact in the refrigerator at 36°F to 46°F (2°C to 8°C). Store prefilled syringes in their original packaging away from light. Do not freeze. Don't shake. Do not expose to high temperatures or direct sunlight.
Allow Voyxactprefilled syringe to come to room temperature77°F (25°C) for 15 to 30 minutes before injection. StoreVoyxactprefilled syringes in the original carton to protect them from light. OnceVOYXACT prefilled syringe has reached room temperature, do not return it to the refrigerator. Do not use ifVoyxacthas been left at room temperature for 7 days or more.
6. Taboo:
Voyxact is contraindicated in patients who are severely allergicsibeprenimab-szsi orany of the excipients of Voyxact.
7. Mechanism of action:
The active ingredient in Voyxact, sibeprenlimab, binds to APRIL with a dissociation constant (KD) of 0.95 pM, blocking the signaling of B cell maturation antigen (BCMA) and transmembrane activator, calcium regulator and cyclophilin ligand interactor (TACI) receptors. Inhibition of APRIL leads to a decrease in serum galactose-deficient immunoglobulin A1 (Gd-IgA1) levels, which is related to the pathogenesis of IgAN.
8. Pharmacodynamics:
In healthy subjects following a single subcutaneous injection of 400 mg of Voyxact, serum levels of APRIL were reduced by more than 90% on day 3, and this reduction persisted through day 42. In a prospective study, VoyxactIgAN patients treated every four weeks experienced APRIL suppression of >90% at week four and continued throughout treatment. Serum Gd-IgA1, IgA, IgG, and IgM levels decreased over 4 weeks, reaching a plateau by week 24, and by week 48, mean serum levels decreased from baseline: Gd-IgA1 decreased by 67%, IgA decreased by 69%, IgG decreased by 35%, and IgM decreased by 75%.
Reference materials:https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=c8a93b32-0676-4704-86dc-cb65f425c6e5
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















