Momelotinib manufacturer and drug source information
Momelotinib is an oral JAK1/JAK2 inhibitor mainly used to treat patients with moderate to severe myelofibrosis. The drug was developed by the US company Gilead Sciences and was initially used in clinical trials and in the treatment of refractory myelofibrosis. As an innovative targeted drug, molotinib has shown the potential to improve splenomegaly, relieve anemia and control systemic symptoms in multiple international clinical trials, providing a new treatment option for patients with moderate to severe myelofibrosis.
In terms of drug marketing and production, molotinib is currently in the clinical marketing or approval stage in some countries and regions. The United States, Europe and other countries have registration and supply of original drugs, but they have not yet been officially launched in China. Therefore, if domestic patients need to use molotinib, they need to obtain the drug through formal overseas channels. These channels often include purchasing prescription drugs from overseas hospitals, international drug delivery services, or participating in clinical trials to obtain drugs.
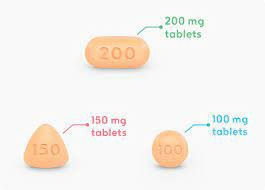
In overseas markets, molotinib original drug is mainly supplied in the form of American version, European version and Turkish version. Different versions have differences in price and supply channels. Patients should pay attention to the legality and formality of drug sources when purchasing and avoid obtaining them through informal channels to ensure drug quality and safety. At the same time, local laws, regulations and transportation requirements must be followed when purchasing to ensure the stability and effectiveness of the medicine during transportation.
In addition, countries such as Laos also have generic drugs approved by regulatory authorities on the market. The ingredients of the drugs are basically the same as the original drugs and the prices are relatively low. This type of generic drugs provides patients with an economically feasible treatment option, but it also requires confirmation of the manufacturer's qualifications and the legality of the drug. In short, molotinib is available from various sources. Domestic patients should choose formal channels to obtain it under the guidance of doctors, and use it rationally in conjunction with clinical medication plans to ensure efficacy and safety.
Reference materials:https://www.drugs.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















