Medical insurance has covered it! Axitinib combined with immunotherapy opens up a new paradigm for kidney cancer treatment
In 2025, at the annual meeting of the American Society of Clinical Oncology (ASCO), the treatment field of renal cell carcinoma (RCC) once again received a major update. A study of axitinib (Axitinib) combined with the PD-1 inhibitor pembrolizumab (Pembro The long-term follow-up study of lizumab) showed that this regimen not only significantly prolonged the overall survival (OS) of patients with advanced renal cancer, but also demonstrated stable and controllable safety in real-world applications. This result once again thrust axitinib into the spotlight of global cancer treatment.
As a second-generation highly selective vascular endothelial growth factor receptor (VEGFR) inhibitor, axitinib has been active in many tumor types such as renal cancer and thyroid cancer in recent years. Especially when combined with immunotherapy, it has become the first-line treatment standard for advanced renal cancer. At the same time, with its full launch and inclusion in medical insurance in China, the accessibility of axitinib has significantly improved, and more and more patients can obtain this innovative targeted therapy at a more reasonable price.
ASCOHeavy data from the annual meeting: axitinib+PD-1Inhibitor therapy refreshes survival record
At the2025ASCO annual meeting, the five-year follow-up data of the KEYNOTE-426 study was announced. This study included patients with advanced clear cell renal cell carcinoma and compared axitinib plus pembrolizumab with conventional sunitinib (Sunitinib) monotherapy.
The key results are as follows:
5Year overall survival rate (OS): the combination group reached 41%, while the control group was only 30%;
The median overall survival time: more than 4 years in the combination group, and about 2.7 years in the control group;
Objective response rate (ORR): the combination group was 60%, which was significantly higher than sunitinib’s 39%;
Complete response rate (CR): The combination group reached 12%, setting a new high for targeted + immunotherapy.
Researchers pointed out that axitinib improves the tumor microenvironment by precisely inhibiting the VEGFR pathway and helps enhance the penetration and effect of immunotherapy, thus playing a key role in the "target-immune combination" model. The results of this study have also been updated by the NCCN and ESMO guidelines, which clearly recommend axitinib + pembrolizumab as the preferred first-line treatment for advanced renal cancer.
VEGFPathway: Axitinib precisely blocks the "vascular supply line" of tumors
Axitinib is a highly selective VEGFR-1, 2, 3 inhibitor. By blocking the binding of VEGF to its receptor, it inhibits the formation of new blood vessels, thereby "starving" tumor cells that rely on blood vessel supply. Compared with early multi-target TKI drugs (such as sunitinib and sorafenib), axitinib has higher selectivity, easier to control side effects, and can achieve more precise anti-angiogenic effects.
Its pharmacological mechanism is mainly reflected in the following three aspects:
1.Inhibit tumor angiogenesis: directly block theVEGFR signaling pathway and limit the supply of nutrients and oxygen;
2.Improve the tumor microenvironment: reduce vascular abnormalities within the tumor and enhance the penetration of immune cells;
3.Collaboration with immunotherapy: improve the immune suppression status of tumors and create a good "combat environment" for PD-1/PD-L1 inhibitors.
This complementary mechanism makes axitinib an irreplaceable "basic partner" in combination immunotherapy.
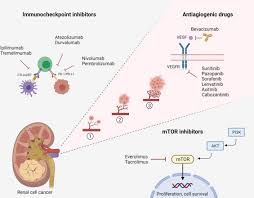
Expansion of indications: From kidney cancer to thyroid cancer, the application landscape continues to expand
1. Advanced renal cell carcinoma (RCC)
Axitinib was approved by the FDA in 2012 for the treatment of patients with advanced RCC who are resistant to previous treatments. In 2019, its combination with pembrolizumab was approved as a first-line treatment, marking the official entry of the "target immunity era" in the treatment of advanced renal cancer.
The National Food and Drug Administration of China (NMPA) has approved the marketing of this combination therapy in 2021, becoming one of the first-line standard treatments recommended by domestic guidelines.
2. Thyroid cancer
Multiple studies have shown that axitinib also has certain efficacy in radioactive iodine-refractory thyroid cancer. Its high target selectivity can reduce the common cardiovascular toxicities of traditional TKIs, allowing it to demonstrate therapeutic potential in partially drug-resistant or relapsed patients.
3. Exploration of other solid tumors
In recent years, axitinib has also been used in the study of combination regimens for multiple tumor types such as liver cancer and lung cancer. For example, the preliminary research results of axitinib + immunotherapy in the second-line treatment of hepatocellular carcinoma showed that the ORR reached more than 25% and was well tolerated, laying the foundation for future expansion of indications.
Side effects and medication precautions
The overall tolerability of axitinib is better than that of earlyTKI drugs, but some manageable adverse reactions may also occur:
1.Common side effects: high blood pressure (about 40%), diarrhea, fatigue, loss of appetite, stomatitis;
2.Laboratory abnormalities: elevated liver enzymes, hypothyroidism;
3.Serious but rare: bleeding, thrombosis, gastrointestinal perforation.
Management suggestions:
1. Blood pressure should be closely monitored in the early stage of medication, and antihypertensive drugs should be given if necessary;
2. Regularly review liver, kidney and thyroid function;
3.When adverse reactions of grade ≥3 occur, suspension or dose reduction may be considered;
4.When combining immunotherapy, you need to be alert to the superimposed risk of immune-related adverse events.
Price information and domestic drug purchasing channels
At present, axitinib has been officially launched in the country and is included in medical insurance.
1. Domestic price: The price of each box of imported original drug (Pfizer) is about more than 4,000 yuan. The actual out-of-pocket amount drops significantly after medical insurance reimbursement. For details, please consult the local hospital pharmacy;
2. Foreign generic drugs: mainly from India and Laos, the price is around a few hundred yuan, and the ingredients are basically the same as the original drugs, but they need to be obtained through formal channels to avoid buying fake drugs or illegal products.
With the improvement of the medical insurance coverage and procurement system, the accessibility of axitinib in China has been greatly improved, and patients no longer need to bear high costs through overseas purchase.
Patient concerns: Who is suitable for axitinib combined with immunotherapy?
Axitinib+PD-1Although combination therapy with inhibitors has significant efficacy, it is not suitable for all patients. Generally recommended groups include:
Intermediate and high risk late stageRCCPatients: IMDCPatients with intermediate and high risk stratification benefit the most from combination therapy;
Patients with good physical status:ECOG 0-1 are better tolerated;
Patients with clearVEGF driving characteristics: the combination regimen can quickly shrink tumors and delay progression;
People who have no contraindications to immunotherapy: such as those without active autoimmune diseases, transplant history, etc.
Exploratory applications for thyroid cancer or other tumor types need to be carried out under the guidance of a doctor.
Future Outlook: Axitinib is expected to enter more combination strategies, and the price may further drop.
The future development trend of axitinib will mainly focus on the following directions:
1. Combination with new generation immunotherapy drugs: such as combination with dual antibodies and cell therapy, it is expected to further break through the resistance bottleneck;
2.Biomarker screening: looking for molecular features that can predict the efficacy of combination therapy to improve precision treatment;
3. Generic drugs and centralized procurement: As more domestic generic drugs are approved, the price of axitinib is expected to drop further, benefiting more patients;
4.Expansion of indications: Clinical research in thyroid cancer, liver cancer and other fields may make new breakthroughs in the next few years.
From the precise inhibition of single-agent targeted therapy to the breakthrough of "target-immune combination" regimen to extend survival, axitinib has witnessed a paradigm shift in renal cancer treatment. Its high selectivity, good tolerability and high synergy with immunotherapy make it the backbone of first-line treatment for advanced RCC. With the advancement of medical insurance coverage and the emergence of generic drugs, axitinib is no longer an "unattainable" targeted drug, but is becoming a standard regimen that more and more patients can actually benefit from.
xa0
References:
1.ASCO 2025Annual Meeting Report:https://meetings.asco.org
2.KEYNOTE-426 Trial - N Engl J Med 2019; 380:1103–1115.
3.NCCN Guidelines Kidney Cancer 2024:https://www.nccn.org
4.ESMO Clinical Practice Guidelines: Renal Cell Carcinoma 2024.
5.National Medical Insurance Catalog (2023 version) drug information disclosure: https://www.nhsa.gov.cn
6.Pfizer Axitinib Product Information:https://www.pfizer.com
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















