How effective is Ivosidenib in treating leukemia and cholangiocarcinoma? A complete explanation of price differences between domestic and foreign countries and ways to purchase medicines
1. Introduction of hot spots: breakthrough in the era of precision medicine
2023At the annual meeting of the American Society of Hematology (ASH), targeted therapy for cancers related to IDH1 gene mutations became a hot topic. Studies have shown that patients using Ivosidenib (Ivosidenib) have achieved unprecedented efficacy in the treatment of acute myeloid leukemia (AML) and cholangiocarcinoma, which has given countless patients and doctors new hope.
In the context of the rapid development of precision medicine, more and more malignant tumors are discovering driver mutations through genetic testing, thus achieving "targeted" targeted therapy . IDH1 mutation is one of the typical examples, and ivonib is the first IDH1 inhibitor approved by FDA.
For many patients diagnosed with AML or cholangiocarcinoma, traditional treatments often have limited efficacy, severe side effects, and high recurrence rates. The advent of ivonib has changed the situation of "lack of effective options" in the past, making it a highly searched keyword by patients and family members.
2. Pharmacological mechanism and indications of ivonib
1. Mechanism of action
Avonib is a selective IDH1 inhibitor, mainly targeting patients with IDH1 gene mutations. IDH1Mutation can cause metabolic abnormalities in tumor cells and produce a carcinogenic metabolite 2-hydroxyglutarate (2-HG), thereby preventing cell differentiation and promoting unlimited proliferation of cancer cells. Ivonib inhibits the activity of mutated IDH1 enzyme, reduces 2-HG levels, and restores the differentiation of cancer cells, thereby achieving the purpose of treatment.
2.Approved indications
Acute myeloid leukemia (AML): used to treat relapsed /refractory (R/R) AML patients, or as monotherapy for patients with newly diagnosed IDH1mutationsAML.
Cholangiocarcinoma (Cholangiocarcinoma): used for patients with IDH1 mutated advanced cholangiocarcinoma who have received at least one systemic therapy in the past.
Other exploratory indications: Some clinical trials are studying its efficacy in glioma and other solid tumors, showing certain potential.
3. Clinical research data: What is the real-world efficacy?
1. AMLTreatment Data
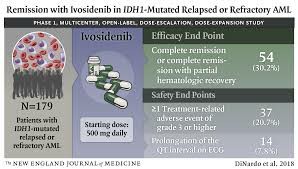
In a pivotalI phase clinical trial, ivonib was used in patients with relapsed/refractoryAML, and the results showed:
Complete response rate (CR): approximately 30%.
Overall response rate (ORR): more than 40%.
Median overall survival (OS): 8.8 months, and some patients exceed 12 months.
Compared with traditional chemotherapy, ivonib is better tolerated and has relatively controllable side effects. It is especially suitable for patients who are elderly or who cannot tolerate intensive chemotherapy.
2. Bile duct cancer treatment data
In the ClarIDHy III phase trial, ivonib significantly improved progression-free survival compared with placebo in patients with IDH1 mutated cholangiocarcinoma:
MedianPFS: 2.7 months vs 1.4 months.
Disease control rate (DCR): more than 50%.
Overall survival (OS): In long-term follow-up, some patients survive more than 1 years.
For patients with cholangiocarcinoma who previously lacked effective second-line treatment options, ivonib has undoubtedly filled the gap.
3. Side Effect Summary
Common adverse reactions include: leukopenia, QT interval prolongation, fatigue, nausea, diarrhea, etc. Overall, most side effects can be managed with dose adjustment and supportive care.
4. The listing and price of avosidenib
1. Domestic listing situation
Currently, ivonib has been approved for marketing in mainland China and is mainly suitable for patients with IDH1 mutations and AML and cholangiocarcinoma.
Price situation: The price of a box of medicine is 3~4 RMB. It is not included in the national medical insurance catalog, so patients need to pay for it at their own expense.
How to purchase medicine: You can purchase it with a prescription at the pharmacies of some large tertiary hospitals.
2. Comparison of foreign markets
Laos generic drugs: The price is about 3000 RMB, the ingredients are the same as the original drugs Basically the same, the price is much lower than that in China.
European and American original drugs: The price is close to that in mainland China, and the monthly cost is often thousands of dollars.
3. Patient selection and financial burden
For domestic patients, the lack of coverage by medical insurance makes the price of avonib extremely high. The cost of a box of 30,000 to 40,000 yuan puts most families under huge financial pressure. In contrast, the low-price advantage of generic drugs in Laos is prominent, but the purchase channels must be formal, otherwise there may be risks of counterfeit drugs.
5. Frequently Asked Questions and Medication Concerns of Patients
Q1: Which patients is Ivonib suitable for?
A: Mainly suitable for patients with IDH1 mutated AML or cholangiocarcinoma. Genetic testing is required to confirm before use.
Q2: How long does it take to take effect?
A
Q3: Is long-term medication safe?
A: Ivonib can be used for a long time, but electrocardiogram, liver and kidney function and blood pictures need to be monitored regularly to prevent serious adverse reactions.
Q4: Can medical insurance reimburse me?
A: It is currently not included in the medical insurance catalog. Patients need to pay the full amount out of their own pocket. Some public welfare assistance projects may provide help.
Q5: Are generic drugs as effective?
A: The ingredients of Lao generic drugs are basically the same as those of the original drugs, but there are slight differences in production processes and regulatory standards. It is recommended that patients purchase it from regular channels and use it under the guidance of a doctor.

6. Real world case sharing
Case1:AMLPatient
A 65 man was unable to tolerate intensive chemotherapy due to poor physical condition. After testing, he was found to have an IDH1 mutation and was prescribed ivonib. After taking the medicine for 2 months, the blood cell indicators gradually returned to normal and the quality of life improved. The treatment has lasted for more than 10 months.
Case2: Patient with cholangiocarcinoma
45 female, received ivonib treatment after failure of second-line chemotherapy, her condition was stable for more than6 months. The main side effects were mild fatigue and elevated blood lipids, which were well controlled after adjusting her lifestyle.
These cases demonstrate that avosidenib provides real and visible benefits to high-risk patient groups.

7. Future Prospects: Application Prospects of Avonib
With the popularity of precision medicine, IDH1 mutation testing will become more and more common. The application scenarios of ivonib may also be expanded from hematological malignancies to more solid tumors.
Future development directions include:
1.Combination therapy: Combined with BCL-2 inhibitors, immunotherapy, etc. to further improve the efficacy.
2.Early application: Explore the application value in patients with newly treated AML or early-stage cholangiocarcinoma.
3.Medical insurance negotiation: If it can be included in the medical insurance directory, it will significantly improve patient accessibility.
8. Summary
Ivosidenib (Ivosidenib), as the world's first approved IDH1 targeted inhibitor, is a milestone in the treatment of AML and cholangiocarcinoma. It significantly improves patient prognosis by precisely inhibiting IDH1 mutations, especially when traditional treatments are ineffective, bringing new hope to patients.
However, although it is currently on the market in mainland China, it is not covered by medical insurance, and the price is as high as 3~4ten thousand yuan/ box, causing a huge burden to the patient's family. In contrast, the price of generic drugs in Laos is only 3,000, which greatly reduces economic pressure, but you need to be cautious when purchasing and be sure to go through formal channels.
With the accumulation of more clinical data and the advancement of medical insurance policies in the future, ivonib is expected to be popularized among a wider range of patient groups. For patients with confirmed IDH1 mutated AML or cholangiocarcinoma, ivosidenib is undoubtedly an important treatment option worthy of attention.
xa0
References
1.Stein EM, et al. Ivosidenib in IDH1-Mutated Relapsed or Refractory AML. NEJM, 2017.
2.Abou-Alfa GK, et al. Ivosidenib in IDH1-Mutant Cholangiocarcinoma (ClarIDHy Trial). Lancet Oncol, 2020.
3.FDA Drug Database – Ivosidenib (Tibsovo) Approval Documents.
4.National Food and Drug Administration (NMPA) drug registration database.
5.Chinese Society of Clinical Oncology (CSCO) Diagnosis and Treatment Guidelines for Cholangiocarcinoma (2023 Edition).
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















