What is the original drug information of enzalutamide/enzalutamide?
Enzalutamide/Enzalutamide (Enzalutamide), as a new type of androgen receptor inhibitor, plays an important role in the treatment of prostate cancer. Its original drug was jointly developed by Astellas Pharma Inc. and Medivation Inc. and was approved for marketing by the US FDA in 2012. In China, the original drug of enzalutamide has been approved for marketing and is reflected in the medical insurance catalog in some regions.
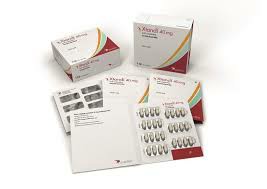
The original drug of enzalutamide exists in the form of soft capsules, with a common specification of 40mg. The drug is sold under the global trade name Xtandi and is also marketed under the trade name "Ancotan" in the Chinese market. The main component of Xtandi is enzalutamide. Its mechanism of action is by inhibiting the nuclear translocation and DNA binding of the androgen receptor, thereby blocking the androgen signaling pathway and inhibiting the proliferation of prostate cancer cells. The drug has shown good efficacy and tolerability in clinical studies and has become one of the important options for prostate cancer treatment.
In China, the original drug of enzalutamide has been approved for marketing by the State Food and Drug Administration and is reflected in the medical insurance catalog in some regions. Patients can use this drug for treatment through medical institutions if they meet the indications. It should be noted that the medical insurance reimbursement policy for enzalutamide may vary by region. Some regions may have additional requirements for patients' age, liver and kidney function and other factors. Therefore, before using enzalutamide, patients should consult their local medical insurance department or medical institution to understand the specific reimbursement conditions and procedures.
As the incidence of prostate cancer increases, the demand for enzalutamide in the Chinese market is gradually increasing. In the future, as more regions include enzalutamide in the medical insurance catalog, its market coverage is expected to further expand. In addition, with the deepening of clinical research, enzalutamide may also play a role in the treatment of other indications, providing patients with more treatment options.
Reference materials:https://www.xtandi.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















