Chemical synthesis route and preparation method of momelotinib
Momelotinib is a new kinase inhibitor that has been widely studied and used in the treatment of myelofibrosis (MF). As a drug in the form of dihydrochloride monohydrate, molotinib is structurally designed to target signaling pathways such as JAK1, JAK2 and ACVR1. Therefore, it can not only improve symptoms related to myelofibrosis, but also effectively alleviate the accompanying anemia problem. Compared with traditional drugs, its clinical advantage lies in taking into account both hematopoietic regulation and symptom control, which is also an important reason why it has received international attention.
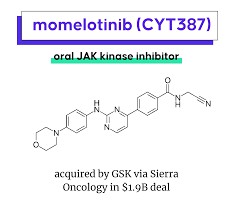
From the chemical structure point of view, the systematic name of molotinib is n-(cyanomethyl)-4-(2-{[4-(morpholin-4-yl)phenyl]amino}pyrimidin-4-yl)benzamide dihydrochloride monohydrate, with a molecular formula of C23H22N6O2·2HCl·H2O and a molecular weight of 505.40. The design of the morpholine group and pyrimidine ring in this structure gives it high selectivity and stability for binding to kinases. As a tablet dosage form, common strengths include 100mg, 150mg and 200mg, allowing for individualized treatment according to the clinical needs of different patients.
In terms of synthetic route, the preparation of molotinib mainly revolves around the pyrimidine core skeleton. The general method is to first synthesize the pyrimidine ring substituent, and then connect it to the benzamide fragment through amidation reaction, and finally obtain the target compound under the conditions of introduction of cyanomethyl and morpholine groups. The entire process often involves nucleophilic substitution, condensation and later hydrochloric acid treatment steps to obtain a stable dihydrochloride crystal form. The advantage of this route is that it can effectively ensure the purity of the intermediate and the crystallization characteristics of the final product, thereby improving the stability and bioavailability of the preparation.
With the optimization of the synthesis process, the scale-up of molotinib in industrial production has gradually matured. Strict control over solvent selection, reaction temperature and purification process during the preparation process ensures the consistency and quality controllability of the drug in large-scale production. Especially in the hydrochloride crystallization stage, by controlling the solvent ratio and temperature gradient, high-purity finished products can be obtained, which is beneficial to the safe application of drugs in clinical applications.
Reference materials:https://en.wikipedia.org/wiki/Momelotinib
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















