Asiminib hydrochloride tablets (Asiminib) are officially launched in China! New options for patients with chronic myelogenous leukemia (CML)
The latest pharmaceutical news express: 2025year5month13day
In an era when targeted therapy drugs are emerging in endlessly, the advent of Aceminib is not just a simple supplement to the launch of a drug, but represents an innovative upgrade in the concept of targeted therapy. Acemini is the first and currently only "STAMP" mechanism (Specifically Targeting the ABL Myristoyl Pocket, a BCR-ABL inhibitor that specifically targets the ABLmyelination pocket), has been included in the recommendations of clinical guidelines for the treatment of CML in many countries around the world.

01 The domestic launch date has been revealed and has not yet been included in the medical insurance catalog
On 2025MayMonth13, the State Food and Drug Administration (NMPA) officially approved Assimini to be marketed in China under the name "Assiminib Hydrochloride Tablets". As a targeted drug with a new mechanism, its launch has aroused great concern among the hematology and oncology departments and CML patient circles.
However, it is reported that asinib hydrochloride tablets have not yet been included in the national medical insurance directory, which means that patients still need to purchase the medicine at their own expense. The specific price varies slightly depending on the hospital, city and supply channel. Patients in need of medicine are recommended to consult the local hospital pharmacy or doctor for the latest quotation.
Original versions of foreign drugs have been on the market in Europe and the United States for many years, and are very expensive, with monthly treatment costs reaching tens of thousands of yuan. We learned from multiple overseas channels that the Lao version of generic drugs is currently on the market, with a price of approximately4000 It is about RMB 10,000, and its ingredients are basically the same as the original drug, making it an affordable alternative for many overseas patients.

02 Aximini: How does the new generation of precision targeting mechanism differ from traditional drugs?
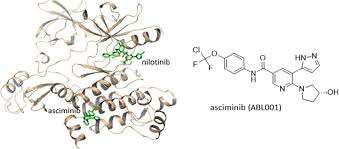
Traditional treatments for chronic myeloid leukemiaTKI drugs, such as imatinib (Imatinib), nilotinib (Nilotinib), dasatinib (Dasatinib pan>), etc., all act on the ATP binding site of BCR-ABL kinase, while Asiminib takes a different approach - acting on the myelination binding pocket of ABL protein (Myristoyl pocket), this site is different from previous TKI drugs and represents a new path in the field of targeted therapy.
Aximinib locks the ABL kinase in an inactive state, thereby achieving a more precise inhibitory effect, especially showing unique advantages in dealing with drug-resistant mutations such as T315I. This type of mutation is one of the most challenging conditions in the treatment of CML, and the mechanism of aceminib provides new treatment hope for such patients.
03 The clinical research data are impressive and highly recommended by international guidelines
Globally, the efficacy of aceminib has been confirmed through multiple clinical studies. Especially inASCEMBL In phase III clinical trials, the study compared the efficacy of aximinib and bosutinib (Bosutinib) in patients with chronic TKI resistance or intolerance CML:
Aximinib-treated patients achieved major molecular response (MMR) at 24 weeks in 25% compared with 13% in the control group with bosutinib.
The efficacy remains more stable, and the incidence of side effects is significantly reduced.
Based on the above results, aceminib has been included in the NCCN guidelines (National Comprehensive Cancer Network) and ELN guidelines ( European Leukemia Network) is recommended as a second-line or multi-line treatment option for CML, especially for patients who are resistant or intolerant to other TKIs.
04 Detailed explanation of the indications and medication groups of Asiminib
The main indications currently approved in China are:
1.For the treatment of TKI chronic stage patients who are ineffective or intolerant to TKICMLadult patients
2.It is also considered a valuable treatment option for T315I mutation-positive CML patients (need to be evaluated on a case-by-case basis)
In terms of usage and dosage, it is recommended according to the instructions:
1.Patients with Philadelphia chromosome-positive chronic myeloid leukemia (Ph+CML) in the chronic phase:
80mg, taken orally, once a day or 40mg, taken orally, 2 times a day
2.Carrying T315I mutation Philadelphia chromosome-positive chronic myeloid leukemia (Ph+CML) Patients in chronic phase:
200 mg, oral, daily2times
Avoid eating for at least 2 hours before and 1 hours after taking this product.
Because it is a drug with an innovative mechanism, patients need to have regular blood routine, liver function and BCR-ABL fusion gene monitoring during the period of taking it in order to evaluate the efficacy and safety.

05 Side effects and medication precautions
Although the side effects of Asiminib are significantly reduced compared to traditionalTKI, the following adverse reactions need to be noted:
1.The most common side effects include fatigue, headache, nausea, diarrhea, rash, etc. Most of them are mild or moderate and can be relieved by themselves or treated symptomatically.
2.Some patients experience cytopenias, elevated liver enzymes or hyperlipidemia, so blood and liver functions need to be monitored regularly during medication.
3.Like other targeted drugs, Aceminib may also interact with other drugs, especially CYP3A metabolism channel drugs (such as antibiotics, antifungals, etc.). It is recommended to use it under the guidance of a doctor.

06 Price comparison of original drugs and generic drugs: How to choose the more cost-effective one?
The domestic version of the original drug has not yet been fully distributed, and the price has not been announced. However, according to previous experience with similar drugs, the monthly treatment cost of the original drug Aceminib may be as high as tens of thousands of yuan. In comparison, a Laotian imitation version of Acemini is currently on the market, priced at approximately 4,000yuan/ box, the drug ingredients are basically the same as the original drugs, which is a realistic choice for many patients to control the cost of treatment.
However, it is worth noting that generic drugs must be obtained through formal and legal channels to ensure drug quality and medication safety, and to avoid uncertain efficacy or safety risks due to unknown channels.

07 Analysis of the prospects of medical insurance: Is it likely to be included in medical insurance?
At present, Aceminib has not been included in the medical insurance catalog, but considering its innovation, clinical urgency and value to drug-resistant patients, the prospect of its inclusion in medical insurance is relatively optimistic. Referring to the path of previous new targeted drugs (such as osimertinib, ivonib, etc.), if aximinib is included in medical insurance in the next few years, it may greatly reduce the financial burden on patients.
Prior to this, it is recommended that patients take the initiative to learn about local major disease reimbursement policies, corporate assistance programs, or participate in clinical studies jointly sponsored by pharmaceutical companies to obtain possible financial assistance.
The launch of Aceminib not only enriches the drug supply, but also improves and breaks through the concept of targeted therapy. As the world's first innovative drug with the "STAMP" mechanism, it provides a practical new solution for those CML patients who suffer from repeated drug resistance or intolerable side effects during TKI treatment.
As it gradually rolls out in domestic hospitals, clinicians gain more experience, and medical insurance policies gradually advance, Assimini is expected to become an important part of the standardized treatment of chronic myelogenous leukemia in the next few years. For patients, rational understanding of drugs and scientific evaluation of treatment paths are the key steps towards long-term remission.
xa0
References:
1.https://www.nmpa.gov.cn/(Official website of the National Medical Products Administration)
2.https://www.nccn.org/guidelines/category_1(NCCN Guidelines)
3.https://www.novartis.com/news/media-releases/asciminib-approved-fda
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















