How to identify the authenticity of Opicapone drugs 2025
Opicapone (Opicapone) is a new generation COMT (catechol-O-methyltransferase) inhibitor, mainly used for Parkinson's disease patients who experience "end-of-dose effect" or "drug efficacy fluctuation" when combined with levodopa treatment. Its mechanism of action is to inhibit the COMT enzyme, delay the metabolism of levodopa in the body, maintain stable blood concentration, thereby prolonging the drug effect time and reducing the "off" time, so that patients can obtain a more stable movement control effect.
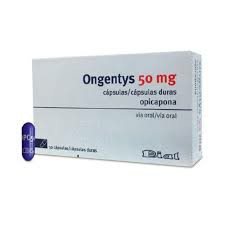
Distinguishing the authenticity of Opicapone drugs is a key link in ensuring the safety and efficacy of medication for patients. The first thing to pay attention to is the outer packaging information of the drug. Genuine drugs should have complete packaging, clear printing, accurate production batch numbers, expiration dates and manufacturer names. When purchasing domestically, there should also be a formal drug approval number and Chinese instructions on the packaging box, which can be checked on the website of the State Food and Drug Administration. If the drug is purchased through a hospital or formal pharmacy channel, it is generally unlikely that counterfeit drugs will appear, but if it is obtained through an unofficial e-commerce platform or purchasing agent channel, the risk increases significantly.
Secondly, the manufacturer of the drug is also an important clue to identify the authenticity. Opicapone was originally developed by BIAL. Its trade name in the European and American markets is usually "ONGENTYS". If the source of the drug is marked as an informal manufacturer, or there is no clear identification of BIAL company information, you need to be vigilant. In addition, observing whether the color, size, and markings of the tablets themselves are consistent can also help identify abnormalities in counterfeit drugs. Genuine products usually have uniform color and texture, while fake drugs may be damaged, have unclear indentations, and have color deviations.
Opicapone has been approved for use in Europe, the United States and some parts of Asia, but it has not yet been fully launched in China. Due to the increasing clinical demand for this drug, generic drugs or counterfeit products are gradually appearing on the market through informal channels. How to identify the authenticity of Opicapone has become an important issue to ensure patient medication safety.
Reference materials:https://www.ongentys.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















