How to use mitomycin intravesical solution (Zusduri) correctly? Detailed explanation of operation process
Before using mitomycin intravesical solution (Zusduri), physicians need to conduct a comprehensive evaluation of the patient, including cystoscopy to confirm the tumor type is "low-grade recurrent intermediate-risk non-muscle-invasive bladder cancer" (LG-IR-NMIBC) and exclude patients with muscle-invasive or high-risk tumors. Routine urine testing should be performed before medication to ensure that there is no serious urinary tract infection or obvious hematuria. Patients should be informed that they need to empty their bladder in advance on the day of infusion and drink less water to ensure that the medication remains in the bladder for sufficient time.
ZusduriBefore use, professional medical personnel will mix and activate the medicinal solution in a sterile environment to form a RTGel® gel dosage form, ensuring that it can slowly solidify and release the medicinal effect at body temperature. The medicinal solution is slowly injected into the bladder through the urinary catheter. The general dose is 215 mg mitomycin dissolved in the RTGel carrier each time. The entire injection process needs to be completed in the outpatient clinic and takes a few minutes. The urinary catheter is removed after use without hospitalization, and the patient can observe and rest at home.
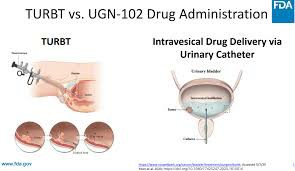
After infusion, it is recommended that the patient keep the drug solution in the bladder for at least 2 hours before urinating. During this period, the patient should avoid strenuous exercise and frequent changes in body position to enhance the efficacy of the drug. Sexual intercourse, strenuous activities and hot spring baths should be avoided within 24 hours after urination to reduce the adverse stimulation of the drug to surrounding tissues. Some patients may experience mild frequent urination or lower abdominal discomfort. This is a common reaction. If symptoms persist, they should report to the doctor in time.
The recommended course of treatment for Zusduri is intravesical instillation once a week, with continuous 6 weeks as a complete course of treatment. During this period, adverse reactions and bladder conditions need to be assessed each time. After completing the treatment, it is recommended to review the cystoscopy within 3 months to evaluate the tumor response, and then perform regular follow-up imaging and microscopy within 6 months to 1 year to monitor for signs of recurrence. For some patients with a high risk of recurrence, doctors may recommend subsequent intensive infusion or extended follow-up periods.
Reference materials:https://www.fda.gov/drugs/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















