Common packaging forms and dosage instructions for giritinib tablets
Gilitinib is an oral small molecule tyrosine kinase inhibitor (TKI), mainly used to treat adult patients with acute myeloid leukemia (AML) with FLT3 mutations, especially for relapsed or refractory patients. As one of the more advanced AML targeted therapies, giritinib works by selectively inhibiting FLT3 (including IT D and TKD mutated forms) and AXL and other tyrosine kinases, thereby blocking the proliferation and survival signals of leukemia cells. With the promotion of clinical use, understanding the common packaging forms and dosage specifications of giritinib is of great significance for patients to use the drug safely and effectively.
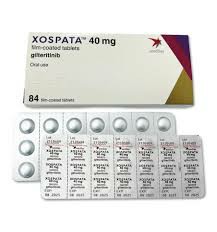
In the international market, the original drug of giritinib is developed and produced by Japan's Astellas Pharma (Astellas Pharma), with the trade name Xospata. According to official information, the common form of the drug is tablets, each containing 40 milligrams (mg). In terms of actual packaging, the common packaging form of giritinib is 40mg tablets, and each bottle contains 90 tablets, that is, a total of 3600mg of active ingredients. This packaging design is set based on the patient's standard daily dosage (120mg). One bottle can just supply a medication cycle of about 28 days, making it easier for doctors and patients to manage the course of treatment.
It is worth noting that the tablets of giritinib are film-coated tablets, light yellow in color, round in shape, and engraved with the logo and dosage identification code of the company "Astellas" on the tablet surface. This dosage form design not only facilitates patient identification, but also effectively protects the stability of the drug ingredients and reduces the degradation of the drug by gastric acid, thereby ensuring the efficacy.
In terms of dosage, the recommended starting dose of gilitinib is 120 mg once daily, and it is usually recommended to take it at a fixed time every day. Patients should swallow the tablets whole and should not crush, chew or break them. Giritinib can be taken on an empty stomach or with food, and there are no special requirements for food. This feature provides patients with greater convenience.
For special patient groups, such as neutropenia, abnormal liver function or abnormal heart rhythm, doctors may adjust the dose or suspend medication based on individual tolerance and laboratory indicators. Clinical data shows that some patients may experience adverse reactions such as increased liver enzymes and prolonged QT interval during treatment. Therefore, regular electrocardiogram and liver function monitoring are necessary. If grade 3 or grade 4 toxicity occurs, medication may need to be suspended and treatment restarted at a lower dose (e.g., 80 mg or 40 mg) upon recovery.
In addition, generic versions of giritinib have also appeared in some generic drug-producing countries, such as India and Bangladesh. Most of these products are also packaged in the form of 40mg tablets, but the specific packaging quantity (such as per box< /span>30 tablets, 60 tablets or 90 tablets) may vary depending on the manufacturer and market demand. The dosage form and packaging design of generic drugs are basically the same as those of the original drugs, and the usage methods are also the same, but patients still need to choose them under the guidance of professional doctors to ensure safety and efficacy.
In general, the packaging format of giritinib is relatively standardized, commonly 40mg tablets, 90 tablets, and three tablets taken once a day (i.e. 120mg) are the standard dosage. This dosing design combines efficacy and safety and has been shown to have good response rates and survival benefits in patients with FL3T mutation-positive AML. Patients should strictly follow the doctor's recommendations during medication and undergo relevant examinations regularly to detect and deal with potential side effects in a timely manner. At the same time, a reasonable grasp of drug dosage forms and packaging information can also help patients avoid risks such as incorrect medication, missed doses, or overdose.
In short, giritinib is an important targeted drug for the current treatment of FLT3mutationsAML. Its dosage form and packaging form are designed with full consideration of patient convenience and treatment continuity. Under standardized medical supervision, mastering dosage and usage specifications will not only help improve the efficacy, but also ensure the safety and compliance of medication to the greatest extent.
Reference materials:https://www.xospata.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















