Ruxolitinib tablets/ruxolitinib manufacturer and original drug
Ruxolitinib tablets/ruxolitinib (JAKAVI) is a selective JAK1 and JAK2 inhibitor, originally developed by the Swiss company Novartis and launched globally under the trade name JAKAVI. In the international market, JAKAVI is an original drug. Its original research and development intention was to target myeloproliferative diseases such as myelofibrosis (MF) and polycythemia vera (PV). With in-depth research, its indications have gradually expanded to rheumatoid arthritis, chronic graft-versus-host disease (cGVHD) after allogeneic hematopoietic stem cell transplantation and other fields. Ruxolitinib reduces the expression of inflammatory factors by inhibiting the JAK-STAT signaling pathway, thereby improving symptoms and controlling the disease.
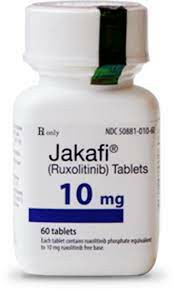
In the Chinese market, ruxolitinib was originally imported by Novartis and officially approved for marketing under the name "ruxolitinib phosphate tablets". It is currently widely used in patients with medium- and high-risk myelofibrosis and polycythemia vera who are ineffective or intolerant to hydroxyurea treatment. According to the adjustment of the national medical insurance drug catalog, this drug has been included in the scope of Class B medical insurance, which greatly reduces the financial burden of patients. Common commercial specifications include 20mg 14 tablets and 5mg 60 tablets.
It is worth mentioning that in addition to the domestic version, some patients are also interested in the original ruxolitinib drug in overseas markets. For example, 5mg*60 tablets of JAKAVI available in Türkiye, the specific price changes with the exchange rate and market supply and demand fluctuations.
As a targeted drug, ruxolitinib's original drug status and production background determine its wide recognition in clinical applications. As an internationally renowned pharmaceutical company, Novartis ensures the quality of drugs and the reliability of clinical research, and also promotes the accessibility of the drug in China. For patients suffering from myeloproliferative diseases, the emergence of ruxolitinib not only provides a new treatment option, but also truly achieves the combination of clinical efficacy and economic affordability due to the support of medical insurance policies.
Reference materials:https://www.jakavi.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















