Is evantumumab already on the market in the country?
Amivantamab is an innovative anti-cancer drug that is an EGFR/cMET bispecific antibody. It is mainly used to treat non-small cell lung cancer (NSCLC), especially those patients who carry EGFR exon 20 insertion mutations. The drug's research and development was led by Johnson & Johnson, and multiple clinical trials have verified its effectiveness and safety in the treatment of specific types of advanced or metastatic non-small cell lung cancer.
Since the approval of evantumumab in the United States in May 2021, the drug has been viewed as an important treatment option, especially for patients whose disease has progressed despite platinum-based chemotherapy. In December 2021, the European Union also granted marketing authorization to the drug, which marked its recognition and use in the international market. The U.S. Food and Drug Administration (FDA) recognized it as a Class I new drug, further confirming its important position in the field of tumor treatment.
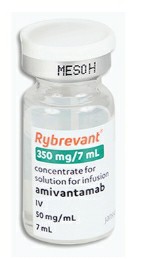
Although evantumumab has been approved in many countries and regions, as of now, according to the latest information, the drug has not yet been officially launched in the Chinese market. This situation may be related to the strict requirements of China's drug regulatory agencies on the new drug approval process. In China, the launch of drugs requires multi-stage clinical trials and evaluations to ensure their safety and effectiveness. In addition, drug market access is also affected by various factors such as production, distribution and pricing policies.
It should be noted that with the continuous research and development of new drugs around the world, the prospect of evantumumab’s launch in China still attracts widespread attention. Many patients and medical professionals hope that this new treatment method can be introduced into the country as soon as possible to meet the growing demand for treatment. However, the exact timing of drug launches remains difficult to predict as it involves complex approval procedures and compliance reviews.
Reference materials:https://www.rybrevant.com/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















