What are the precautions and contraindications when using mitomycin intravesical solution (Zusduri)?
Mitomycin intravesical solution (Zusduri) is an innovative local chemotherapy drug developed by UroGen Pharma. It is mainly used to treat recurrent low-grade intermediate-risk non-muscle-invasive bladder cancer (LG-IR-NMIBC). The drug uses RTGel®thermosensitive gel sustained-release technology to stably release mitomycin into the bladder cavity, replacing repeated surgeries (TURBT) to control tumor progression. Although Zusduri has good efficacy and the advantages of topical application, patients still need to pay attention to a number of precautions and contraindications during use to ensure the safety and effectiveness of the treatment.
1. Assessment and preparation before medication
Before using Zusduri, doctors need to conduct a detailed condition assessment, including pathological diagnosis, imaging examination and urinary system function testing. The applicable population is adult patients diagnosed with "low-grade intermediate-risk non-muscle-invasive bladder cancer", and the tumor is recurrent, non-primary, the lesions do not involve the bladder muscle layer, and there is no evidence of upper urinary tract involvement. This drug is prohibited from being used in patients with muscle-invasive bladder cancer (MIBC), high-grade lesions, bladder carcinoma in situ (CIS), or patients with significant urinary tract obstruction.
In addition, routine urine examination is required before taking the drug to rule out urinary tract infection or significant hematuria. If there is active cystitis or other urinary system inflammation, anti-infective treatment should be carried out first, and the infusion treatment can be started after the infection is controlled.
2. Precautions during medication operation
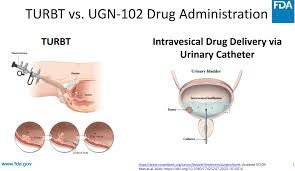
ZusduriIt must be performed by a professional urologist or a trained medical staff, and strict aseptic technique must be followed. The drug is slowly instilled into the bladder through the urinary catheter. The patient should empty the bladder as much as possible before instillation and limit the amount of water consumed within one hour before taking the drug to extend the residence time of the drug.
After the infusion is completed, the patient should be advised to stay supine or move as little as possible, and wait at least 2 hours before urinating. If you experience discomfort or severe abdominal pain during this period, you should contact your doctor in time. To prevent the liquid from irritating the perineal or vulvar skin, rinse thoroughly with clean water after urination.
During the entire treatment cycle (the standard is once a week, a total of 6 times), patients need to maintain close communication with the medical team and report any discomfort or abnormal symptoms, including frequent urination, urgency, hematuria, fever, etc. For moderate or above adverse reactions, it is necessary to evaluate whether to suspend treatment or extend the infusion interval.
3. Taboos and restrictions for use by special groups of people
1. Contraindicated during pregnancy and lactation
Mitomycin, the active ingredient in Zusduri, is cytotoxic and may cause harm to the fetus or infant. There is currently insufficient human research data to confirm its safety during pregnancy and lactation, so its use by pregnant women, those planning pregnancy, and lactating women is strictly prohibited. Women of childbearing age should take reliable contraceptive measures during use, and male patients are also advised to avoid impregnating their partners during treatment.
2. Use with caution in patients with severe bladder dysfunction
If the patient has significantly reduced bladder capacity, urinary dysfunction (such as neurogenic bladder), or difficulty urinating due to severe prostatic hyperplasia, it may not be possible to effectively complete the infusion of the medicinal solution or extend the retention time of the medicinal solution, which will affect the efficacy and increase the risk of adverse reactions. Professional evaluation is required to determine whether it is suitable.
3. Use with caution in patients with severe systemic diseases
If the patient suffers from severe renal insufficiency, abnormal liver function or immune system disease, although Zusduri is a topical drug, it may still produce a certain amount of systemic absorption, leading to systemic toxic reactions. This group of people needs to be closely monitored and evaluated before taking the drug. If the risk is too high, use should be suspended.
4. Potential adverse reactions and preventive measures
UsingZusdurimay cause the following side effects:
Urinary system irritation symptoms: such as frequent urination, urgency, difficulty urinating, bladder discomfort, etc. Most are mild to moderate and usually do not affect treatment.
Hematuria: About some patients will develop hematuria after infusion. In severe cases, the medication should be suspended to find out the cause.
Bladder spasm or pain: Individual patients may experience transient bladder spasm, and analgesics or anticholinergic drugs can be used prophylactically during infusion.
General malaise: A few patients may experience mild systemic reactions such as low fever and fatigue. It is recommended to drink more water and pay attention to rest.
In order to reduce the incidence of adverse reactions, it is recommended to maintain good hydration during the medication cycle, eat reasonably, and avoid irritation from tobacco and alcohol; at the same time, follow the doctor's instructions to regularly check urine, blood routine and renal function indicators.
5. Combination therapy and drug interactions
Currently, Zusduri is mostly a single-agent local treatment. There is insufficient data to support its safety and efficacy in combination with other chemotherapy agents, immunotherapies, or targeted agents. Therefore, during treatment with Zusduri, it is not recommended to use it alternately with other intravesical drugs (such as BCG, epirubicin, etc.) to avoid increased toxicity or local damage caused by drug superposition.
If the patient is receiving other oral or intravenous anti-cancer drugs at the same time, he should explain it to his doctor to avoid uncontrollable risks caused by potential drug interactions.
6. Follow-up management and efficacy evaluation
After completing 6 weeks of standard treatment, patients should undergo follow-up cystoscopy at 12 weeks (i.e. approximately 3 months after treatment) to evaluate whether complete remission (CR) has been achieved. Thereafter, a review will be conducted every 3 to 6 months to monitor recurrence. If recurrence occurs after treatment, re-perfusion, surgery or other treatments can be chosen according to the doctor's advice.
During the entire treatment period, patients should establish a complete disease course record, including each medication reaction, adverse events, review results, etc., to provide a reference for the optimization of future individualized treatment plans.
The launch of Zusduri provides the possibility of treatment that does not rely on surgery for some bladder cancer patients, but it is not suitable for all people. Scientific and reasonable medication evaluation, adequate preoperative preparation, standardized perfusion operations, timely side effect intervention, and strict screening of contraindications are the keys to ensuring the safety and efficacy of treatment. Patients should use this drug under the guidance of a professional urologist and maintain active cooperation to ensure smooth treatment. With the accumulation of clinical experience and the support of more data, Zusduri is expected to play a greater role in the treatment of localized bladder cancer.
Reference materials:https://www.fda.gov/drugs/
[ 免责声明 ] 本页面内容来自公开渠道(如FDA官网、Drugs官网、原研药厂官网等),仅供持有医疗专业资质的人员用于医学药学研究参考,不构成任何治疗建议或药品推荐。所涉药品可能未在中国大陆获批上市,不适用于中国境内销售和使用。如需治疗,请咨询正规医疗机构。本站不提供药品销售或代购服务。
.jpeg)


















